Alzheimer's Disease
Preclinical Research
The primary emphasis of our preclinical research is to understand how the disease operates— from the mechanism of damage to repair. Major efforts are in developing early diagnostic strategies and disease-modifying therapies. These research projects include the study of:
- Disease in the eye’s retina
- Retinal biomarker imaging: can Alzheimer’s be detected noninvasively and definitively with retinal imaging?
- Visual deficits in animal models of aging and AD
- Disease pathogenesis in the brain, retina and body fluids:
- Neuro-immune interactions
- Neurovascular unit
- Infectious etiology
- Epigenome mechanisms and related interventions
- Immune-based therapies
- Blood monocytes in repair and regeneration of central nervous system and cognitive preservation
- Enhancing angiotensin-converting enzyme in immune cells
- Other strategies to preserve brain cells, structure and connectivity, protecting from cognitive decline
- Regenerative medicine

Researchers are refining a noninvasive, in vivo optical imaging test to monitor discrete plaque dynamic appearance and clearance in the retina. The test has proven successful in detecting early signs of Alzheimer's in preclinical experiments and investigative devices in humans. The ability to identify Alzheimer's in human retinas has the potential to revolutionize how we detect early signs of the disease. Our studies also suggest using retinal imaging alongside immune-based therapies to determine which interventions increase the likelihood of significantly retarding and treating Alzheimer's.toxic forms of Aβ and resist its detrimental effects.
- Principal Investigator: Maya Koronyo-Hamaoui, PhD, head of Koronyo-Hamaoui Lab
- Key Research Associate: Yosef Koronyo, MS, LLB
- Key Investigator: Keith L. Black, MD, the Ruth and Lawrence Harvey Chair in Neuroscience, and Chair, Department of Neurosurgery
The continued mission of the Koronyo-Hamaoui Laboratory is to identify pathological hallmarks and associated inflammation, vascular abnormalities and degeneration in the retina of mild cognitively impaired and AD patients. The team investigates how early AD signs appear during disease progression, determine their geometric and layer distribution, their specificity to AD compared with other neurodegenerative diseases and establish the feasibility to serve as biomarkers to predict brain pathology and cognitive status.
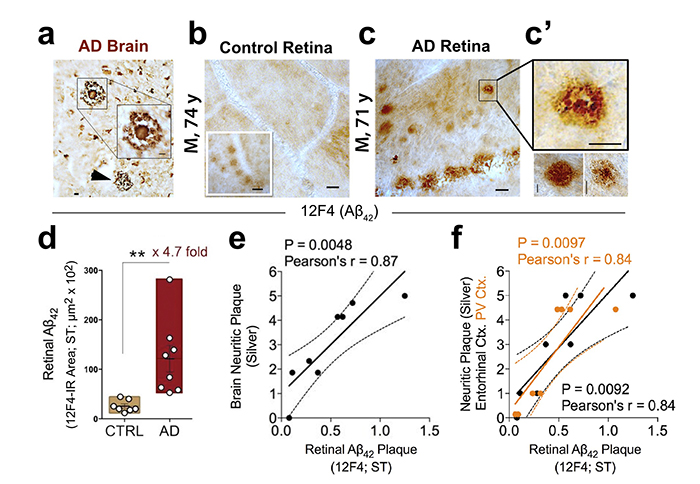
Figure 1. Retinal amyloid pathology in AD patients. Increased retinal Aβ42 deposition correlates with cerebral amyloid plaque burden in Alzheimer’s patients. From the Koronyo-Hamaoui Lab: Koronyo, et al., JCI Insight. 2017.
In the realm of therapy, the Koronyo-Hamaoui Lab studies the therapeutic potential of monocytes and macrophages derived from bone marrow of young donor mice, either activated by glatiramer acetate (GA, also Copaxone) or genetically modified by ACE (ACE10 model), to resist retinal and brain pathology and cognitive decline related to AD in transgenic murine models. The lab continues to investigate the complex relationship between the peripheral immune system and the central nervous system, related to both detrimental and protective outcomes.
The group evaluates the effect of GA immunomodulation in vivo and in vitro and further explores mechanisms of action using advanced high-throughput technologies (proteomics, transcriptomics). In addition, the Koronyo-Hamaoui Lab discovered that GA stimulates OPN/SPP1 in cerebral-recruited macrophages, which resulted in synaptic protection and immune-dependent resilience to AD pathology.
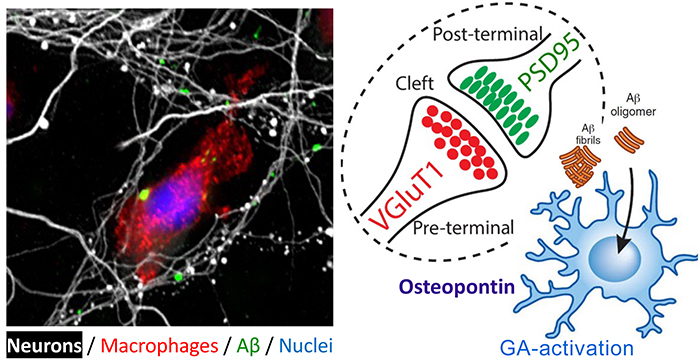
Figure 2. Synaptic and neurite preservation from toxic Aβ1-42 oligomers obtained by GA-activated monocyte-derived macrophages. Red: a macrophage expressing high levels of scavenger receptor; Green: Aβ1-42 oligomers; White: Neuronal processes (in primary neuronal culture). From the Koronyo-Hamaoui Lab: Li, et al., Front. Immunol. 2020.
- Principal Investigator: Maya Koronyo-Hamaoui, PhD
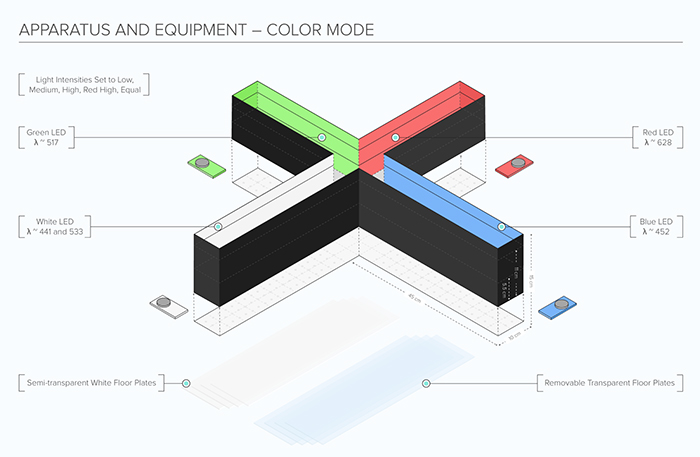
Figure 3. Koronyo-Hamaoui's novel visual stimuli 4-arm maze (ViS4M; also called x maze) to assess color and contrast vision in animal models of aging and Alzheimer's disease. Illustration courtesy of ConductScience/MazeEngineers (2021); From Koronyo-Hamaoui Lab: Vit, et al., Sci Rep. 2021.
Disease pathogenesis in the brain, retina and body fluids:
- Neuro-immune interactions (PIs: Koronyo-Hamaoui, Goodridge, Bernstein)
- Retinal Neurovascular unit (PIs: Koronyo-Hamaoui)
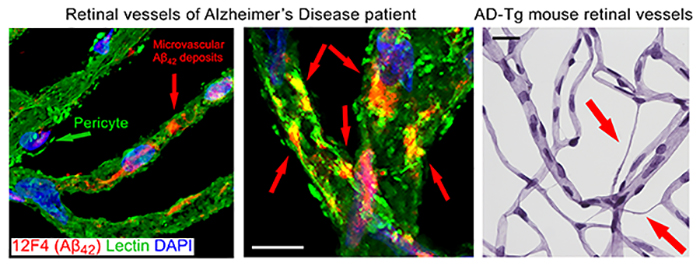
Figure 4. Retinal vasculopathy in Alzheimer's disease. From Koronyo-Hamaoui Lab: Left image: Shi, et al., Acta Neuropathologica. 2020. Right image: Shi, et al., ANP Commun. 2020.
- Infectious etiology (PIs: Koronyo-Hamaoui, Crother, Arditi)
- Epigenome mechanisms and related interventions (PIs: Koronyo-Hamaoui, Black)
- Immune-based therapies (PIs: Koronyo-Hamaoui, Black)
- Blood monocytes in repair and regeneration of central nervous system and cognitive preservation (PI: Koronyo-Hamaoui)
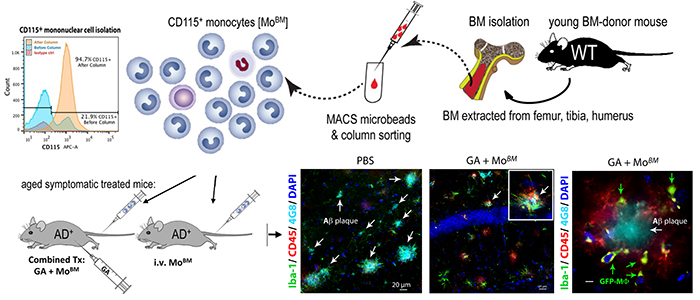
Figure 5. Therapeutic effects of infusing bone marrow-derived monocytes from a young healthy mouse to an Alzheimer's-model mouse, mitigation of brain pathology and restoration of cognitive abilities. From Koronyo-Hamaoui Lab: Koronyo et al., Brain 2015
- Enhancing angiotensin-converting enzyme in immune cells (PIs: Koronyo-Hamaoui, Bernstein)
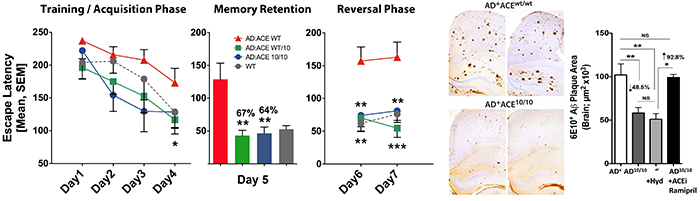
Figure 6. Enhanced capabilities of ACE-overexpressing monocytes and macrophages to curb Alzheimer's pathology and restore cognitive function. From Koronyo-Hamaoui Lab: Bernstein, et al., JCI.2014.
The Koronyo-Hamaoui Lab in collaboration with the Bernstein Lab continue to investigate how targeting overexpression of angiotensin-coverting enzymes (ACE) by macrophages can change the course of Alzheimer's progression. In experimental models, ACE has been shown to be critical in destroying soluble levels of pathogenic Aβ1-42, altering inflammatory responses and preventing cognitive decline.
The research team seeks to determine if ACE can help immune cells eradicate and resist damaging forms of Aβ.
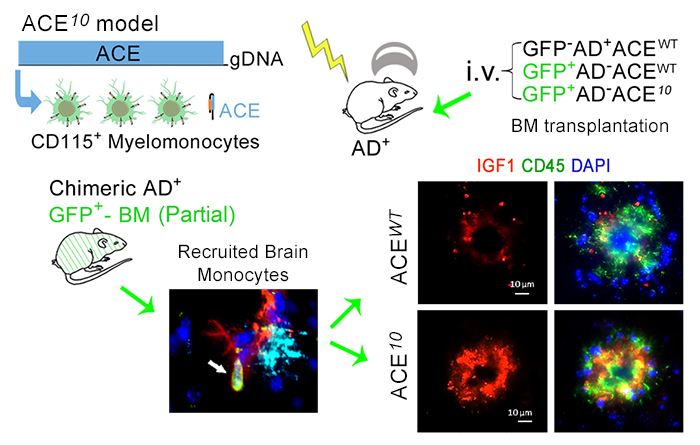
Figure 7. BM transplantation or Blood Infusion with ACE-overexpressing (ACE10) monocytes were found to be highly neuroprotective and prevent cognitive loss in Alzheimer's-model mice. From: Koronyo-Hamaoui,et al., Brain. 2020.
- Principal Investigators: Maya Koronyo-Hamaoui, PhD, and Kenneth Bernstein, MD
We are studying abnormalities in the function of monocytes and macrophages—two types of immune system cells capable of removing amyloid beta (Aβ). Our researchers hypothesize that these cells do not function efficiently in Alzheimer's patients, allowing amyloid accumulation, eventual nerve cell loss and cognitive deterioration. We generate a supply of monocytes and macrophages from patients to study their cells' ability to clear toxic forms of Aβ and resist its detrimental effects.
- Principal Investigator: Helen Goodridge, PhD
The Rutishauser Lab is a systems neuroscience laboratory that uses a combination of in vivo single-unit electrophysiology, intracranial electrocorticography, eye tracking, behavior, and computational and theoretical approaches. The lab helped pioneer the technique of human single-neuron recordings and continues to advance the tools, methods and surgical techniques that allow such experiments. Recent work has focused on single-trial learning, the theta rhythm, faces, error monitoring and memory retrieval.
As part of our cognitive neuroscience program, we are studying how memories are encoded, consolidated, retrieved, and used for making decisions in the human brain. Our aim is to develop a deep mechanistic understanding of human memory processes and to determine how specific aspects of these processes are affected by Alzheimer’s Disease, with the ultimate goal of using this knowledge to develop new treatments for the cognitive effects of Alzheimer’s.
We use a combination of in vivo single-unit electrophysiology, intracranial electrocorticography, eye tracking, behavior, and computational and theoretical approaches. A key approach that we have helped pioneer is the technique of human single-neuron recordings. We continue to advance the tools, methods and surgical techniques that allow such experiments. Lab link: rutishauserlab.org
- Principle Investigator: Ueli Rutishauser, PhD
We collaborate with:
- Department of Pathology and Laboratory Medicine
- Department of Neurology
- Keck School of Medicine of USC
We also collaborate with USC-ADRC and Doheny Eye Institute (UCLA), UCLA's Departments of Neuropathology, Neurology, Molecular Biology Institute and Brain Research Institute, Biopolymer Laboratory and Mary S. Easton Center for Alzheimer's Disease Research.
Clinical Research
The Jona Goldrich Center for Alzheimer’s and Memory Disorders brings the discoveries of Cedars-Sinai basic science, preclinical, translational and clinical researchers on Alzheimer’s disease and related dementias (ADRD) into the best possible care for persons living with dementia.
Utilizing a retinal imaging device for early diagnosis
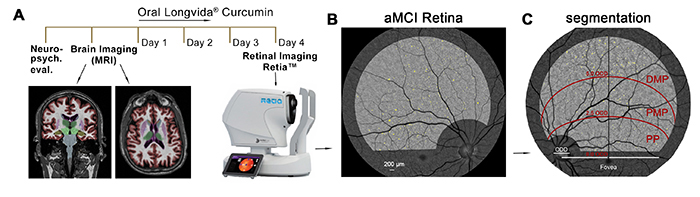
Figure 8. Retinal amyloid imaging in MCI and AD patients was developed by the Koronyo-Hamaoui Lab and NeuroVision Imaging. Adopted from: Dumitrascu, et al., Alzheimer & Demen. (Amst.) 2020.
- Principal Investigator: Maya Koronyo-Hamaoui, PhD.
- Key Investigators: Keith L. Black, MD, and Yosef Koronyo, MS, LLB
Intervention clinical trials: Immune-based therapies and other strategies
The medical care for persons living with Alzheimer’s disease and other forms of dementia is complex and involves a team of health providers and transitions between different sites of care. Our health services research explores innovative ways to achieve the Institute for Healthcare Improvement (IHI) triple aim of better patient outcomes (population health), better experience of care (patient satisfaction) and better value of care. Areas of active investigation include interventions to improve identification of persons with dementia in the clinic, emergency department and the hospital, improving quality and efficiency of dementia care and reduction of rehospitalization rates for hospitalized persons living with dementia.
The Memory & Aging Program is a comprehensive population health approach to the care of persons living with dementia. The interventions span the early identification and intervention of persons-at-risk through health screening to the expert management of challenges associated with moderate to late-stage disease. The novel CEDARS-6 tool enables a personalized care path for persons living with dementia, their families/caregivers and health providers. We are exploring the acceptability and effectiveness of this approach to Alzheimer’s disease health delivery in achieving the triple aim.
With the recent discovery that the disease process for Alzheimer’s disease starts as early as 20 years before the appearance of the first symptom, the Centers for Disease Control and Prevention (CDC), the Alzheimer’s Association and the National Institutes of Health (NIH) have identified Alzheimer’s prevention and risk reduction to be an important area of dementia research. We are investigating the role of modifiable lifestyle factors including cardiovascular risk (blood pressure, diabetes, smoking), physical activity, nutrition, social interactions and sleep in mitigating the risk for developing Alzheimer’s disease.
Are you interested in participating in brain research? We are looking for people who want to contribute to our understanding of brain health, aging and neurodegenerative conditions like Alzheimer’s disease.
Clinical Trials
Clinical trials of investigational compounds are crucial to finding medications to treat Alzheimer’s disease and other neurodegenerative conditions that cause dementia. Participation in a trial is an option for people diagnosed with mild cognitive impairment (MCI) or dementia, or people who are at risk for developing MCI or dementia due to ADRD.
To find out more information about our clinical trials and how to participate, click here.
