Research Areas
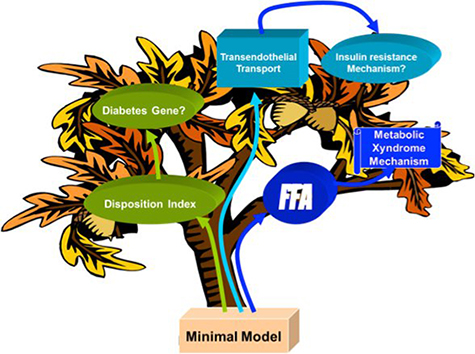
We have discovered that free fatty acids, common lipid molecules in blood, change with time. We reported in animals that FFAs are elevated in the middle of the night (3 am), a result that has been replicated in human volunteers. We have hypothesized that this nocturnal surge in FFA has important metabolic consequences: that is, not only does it contribute to glucose intolerance, but the surge is also responsible for the increase in insulin secretory function that normally accompanies insulin resistance. It is the failure of the pancreatic islets (i.e., beta-cells) to compensate for insulin resistance that is responsible for the prediabetic state. We are examining the explicit role of the FFA surge in the pathogenesis of Type 2 diabetes.
We are also studying the role of sleep in the development of diabetes. We have demonstrated that a single night of sleep interruption is enough to cause insulin resistance equivalent to what is caused by moderate obesity. The mechanisms by which disruption of sleep induce insulin resistance remain to be delineated.
In addition, we are examining the relationship between insulin resistance and cardiovascular disease. In particular, we are interested in the regulation of insulin movement between the bloodstream and tissues that respond to insulin – across the capillary endothelium. We have demonstrated that this transport accounts for the delay in insulin action in vivo, and that fat-feeding induced adiposity can alter this transport rate and cause insulin resistance. We are examining this transport process as a possible target for treatment of insulin resistance in prediabetes and diabetes. We are also studying the ameliorating effect of renal denervation on this transport rate, and on insulin resistance.
In other studies, we are examining the mechanisms by which the antipsychotic drugs can cause increased obesity and Type 2 diabetes.
We continue to use mathematical modeling to develop methods that can be used to provide an efficient picture of metabolic function in single individuals, and to examine the longitudinal changes and genetic control of metabolic function in humans of varying ethnicity.
Contact the Bergman Lab
8730 Alden Drive.
Thalians Building, Room E104
Los Angeles, CA 90048
