Research Areas
Delayed wound healing, erosions, and keratitis are among corneal complications of diabetes that are treated only symptomatically. We have uncovered many markers altered in diabetic corneas. Using adenoviral gene therapy, epigenetic modulation, and novel nanodrugs we corrected aberrant wound healing and marker protein abnormalities in human organ-cultured diabetic corneas. We discovered stem cell dysfunction in diabetic corneas supports that may be the basis of delayed diabetic wound healing.
Current work is focused on normalizing cultured progenitor cells for future transplantation to diabetic corneas to translate novel therapies to clinical application. We are also developing combination nano therapies for glioblastoma targeting immune cells and extracellular matrix in tumor microenvironment.
Gene and Epigenetic Therapy for Diabetic Corneal Stem Cells
The Ljubimov Lab has uncovered a number of markers altered in diabetic corneas. Researchers in the Ljubimov Lab have developed efficient adenoviral gene therapy, influencing the expression of these corneal marker proteins in order to normalize stem cell functions and wound healing in diabetic human corneas in organ and cell culture.
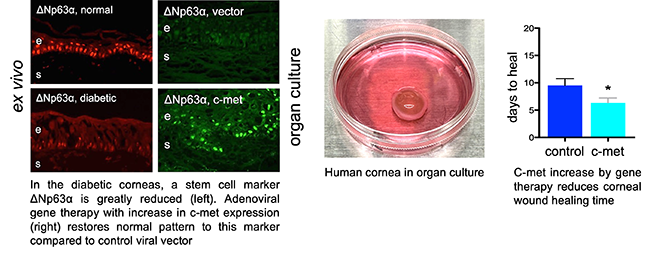
C-met, an oncogene and growth factor receptor, is reduced in diabetic cornea. Adenoviral gene therapy with c-met overexpression can normalize stem cell markers and wound healing in organ cultures of human diabetic corneas.
More recently, we successfully used nontoxic nanopolymers based on polymalic acid for the same task. (supported by NIH grant) We were able to correct aberrant wound healing and abnormal marker protein expression in human organ-cultured diabetic corneas (Fig. 2) using epigenetic mediators including microRNA and DNA demethylator, zebularine (Fig. 3).
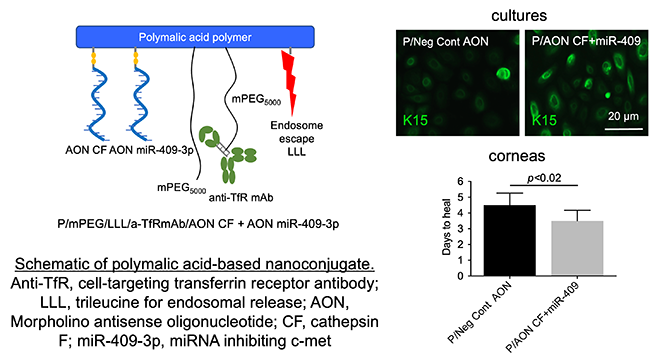
Fig. 2 A polymeric non-toxic nanoconstruct (left) inhibits diabetes-induced cathepsin F and increases diabetes-suppressed c-Met using antisense technology. As a result, stem cell marker keratin 15 expression is normalized (top right) and corneal wound healing is accelerated (bottom right).
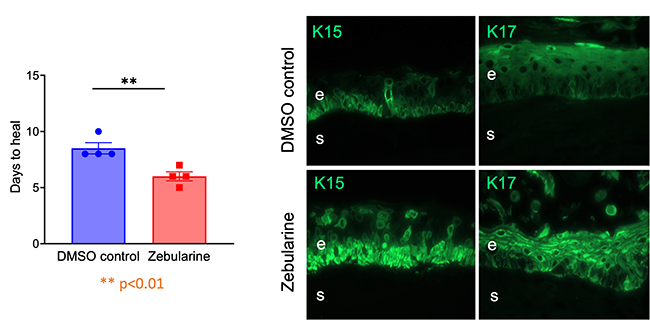
Fig. 3 DNA demethylator Zebularine improves wound healing (left) and limbal stem cell phenotype (by expression of keratins 15 and 17) in diabetic organ-cultured corneas.
Limbal Stem Cell Deficiency
Limbal stem cell deficiency is a consequence of genetic diseases, chronic inflammation and eye burns. It may result in vision loss due to conjunctival ingrowth with neovascularization and is hard to cure even with corneal transplantation. The Ljubimov Lab has generated induced pluripotent stem cells (iPSC) from corneal epithelial cultures. With NIH grant support, an optimized protocol is being developed to differentiate these iPSCs into corneal cells. This protocol would enable the creation of a bankable source of corneal epithelial cells for future transplantation to patients. Our available data suggest that making corneal cells from iPSC may normalize them for future transplantations back to the patient’s eye (see image below).
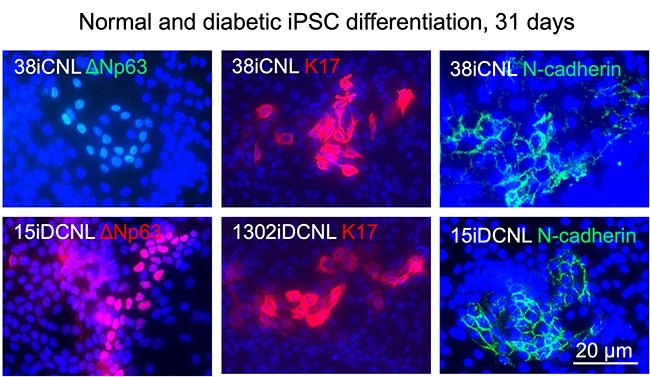
Fig. 4 Corneal epithelial stem cell marker patterns show that cells differentiate to the right lineage. Importantly, diabetic (bottom) and normal cells (top) are very similar by marker expression indicating possible normalization.
Nano Immuno Polymers for Tumor-Specific Targeting and Treatment of Glioblastoma
We are developing nanopolymers based on polymalic acid that can cross blood-brain barrier and inhibit glioblastoma growth in mice. They also can stimulate innate brain immune system by inhibiting glioma marker laminin-411 (it acts through the Notch system). Additional adaptive immune stimulation is provided by nanoconjugate delivering Treg inhibitor anti-PD-1
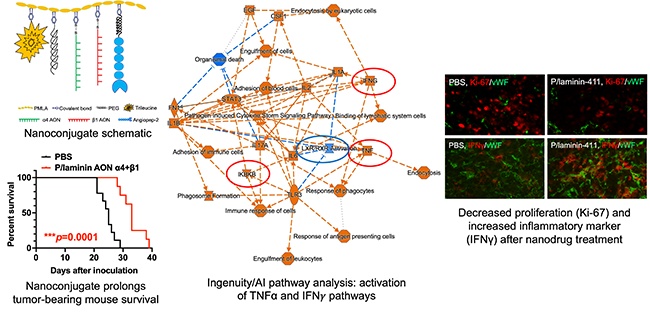
Fig. 5 Top left, schematic of anti-laminin nanoconjugate. Bottom left, this nanoconjugate prolongs mouse survival after intravenous treatment. Center, pathway analysis of treated tumors from RNA-seq reveals activation of inflammatory pathways (IFNg and TNFa) and reduced proliferation. Right, RNA-seq validation by immunostaining.