Research Areas
The Pandol Lab in the Cedars-Sinai Pancreatic Research Program is concentrating efforts to better understand the molecular mechanisms of pancreatic diseases and develop new prevention, diagnostic and effective treatments for these diseases. The Pancreatic Research Program's multidisciplinary team consists of molecular biologists, cell biologists, computational chemists, population scientists, clinical scientists and physicians, all working toward the common goal of enhanced treatments and outcomes for pancreatic cancer, pancreatitis and diabetes patients. Our research is focused on the following:
- Early diagnosis and disease prevention
- Signaling mechanisms, underlying risk factors and disease pathophysiology
- Development of novel therapeutics and enhanced treatments
- Human testing of new treatments
Basic Research
For pancreatic cancer, the Pandol Lab focuses on the mechanisms of the pro-cancer functions of cells in the microenvironment of the cancer (i.e., stellate cells, macrophages and immune cells) that are involved in promoting the growth and metastasis of the cancer. Our aim is to identify specific pathways these cells use to promote the cancer. In addition to developing novel treatments for the cancer, our goal is to prevent occurrence initially and recurrence after initial treatment.
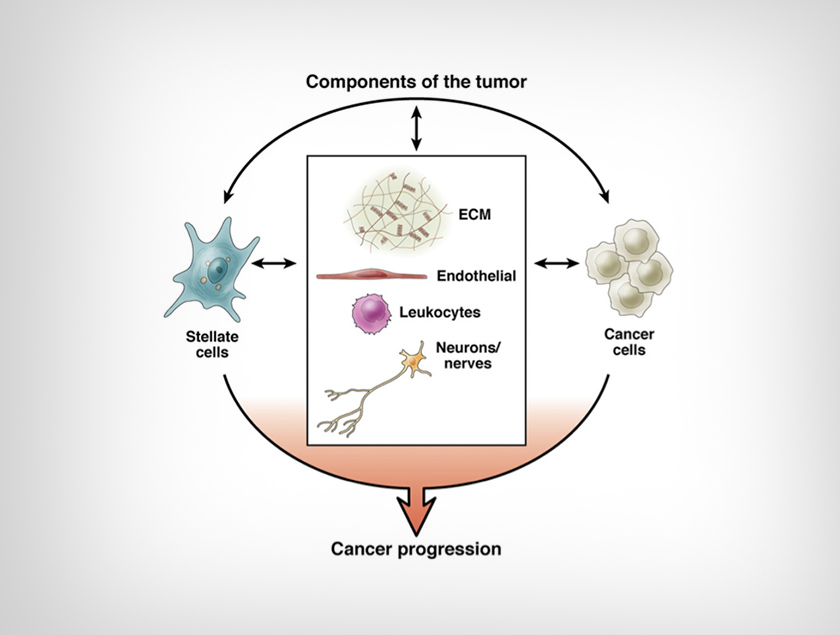
Figure 1: Components of pancreatic cancer. The components of pancreatic ductal adenocarcinoma and potential interactions between the stroma and cancer cells should be considered in developing therapeutic approaches for pancreatic cancer. We are working on the importance of the interactions between cells of the tumor in the growth and metastasis of the disease. Our focus is to use this information to develop preventive strategies and treatments for pancreatic cancer.
One hypothesis driving our research in pancreatic cancer and pancreatitis is that the pancreatic acinar cell and its necessary function — the production and delivery of digestive enzymes — can be stopped at the level of the endoplasmic reticulum (ER). See Figure 3.
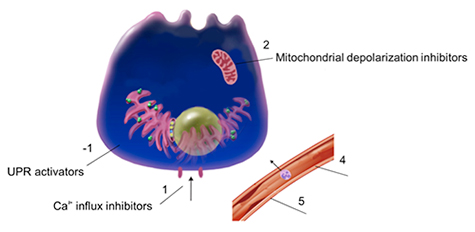
Figure 2: Potential targets for development of therapeutics for acute pancreatitis. This illustration of a pancreatic acinar cell and blood vessel shows therapeutic targets that provide potential treatment opportunities. We are working on developing agents that work on one or more of these targets. Our results are leading to clinical trials for these agents that could show benefit for treatment of pancreatitis.
For diabetes, we focus on how nutrients in the intestine activate gut endocrine cells (i.e., glucagon-like peptide-1 cells and cholecystokinin cells) to regulate glucose metabolism and food intake. The work involves identification of sensors and their functions on the gut endocrine cells with the aim of developing treatments for diabetes through agents that modulate these sensors.
The L cells are interspersed among the intestinal epithelial cells of the intestine. They respond to luminal signals such as fatty acids and bitter molecules, resulting in release of the hormones glucagon-like peptide 1 (GLP-1) and peptide YY (PYY) from their basal surface. GLP-1 and PYY promote insulin secretion, slow gastric emptying and decrease appetite. We are developing agents that interact with the luminal facing sensors to promote release of GLP-1 and PYY to treat diabetes. Our goal is to have agents that can be used for treatment of diabetes.
Clinical Research
Gemcitabine chemotherapy and radiotherapy (RT), given concurrently, remain mainstay treatments for patients with locally advanced pancreatic cancer (LAPC), which cannot be surgically removed and has not spread to other areas of the body. Both gemcitabine and RT kill tumor cells by interfering with their DNA, the genetic blueprint. Veliparib is a poly (ADP-ribose) polymerase (PARP) 1/2 inhibitor; PARP1/2 proteins are also involved in DNA repair. Our work in the laboratory has shown excellent synergy between veliparib (a PARP inhibitor), RT and gemcitabine in killing pancreatic cancer cells grown in culture, and in prolonging survival in mice with pancreatic tumors. Based on these promising results, we are conducting a first-in-human investigational clinical study (Phase I) to test the safety and efficacy of veliparib in combination with gemcitabine and RT for patients with LAPC.
In addition, promising new treatments originating from basic research projects for diabetes, pancreatitis and pancreatic cancer will be considered for Phase I clinical trials after demonstrated in vivo efficacy and safety in animals in the next few years at Cedars-Sinai.
Contact the Pandol Lab
110 N. George Burns Rd.
Davis Building, Room 3096
Los Angeles, CA 90048