Research Areas
The Gibb Lab studies the role of inflammation and innate immunity in detrimental antibody responses to red blood cell (RBC) antigens during blood transfusion. These alloantibodies can cause potentially fatal transfusion reactions and severely limit the availability of RBCs for patients who depend on transfusion. While these responses occur in 3%-5% of all hospitalized patients, patients with some inflammatory diseases, including systemic lupus erythematosus (SLE) and sickle cell disease (SCD), have alloimmunization frequencies as high as 50%. Identifying mechanisms underlying these high rates of alloimmunization could lead to target therapies that prevent anti-RBC antibody responses prior to transfusion, ultimately improving transfusion safety.
Inflammation-induced RBC alloimmunization in lupus
Approximately 160,000 to 322,000 Americans have systemic lupus erythematosus (SLE), which causes end-organ disease, including respiratory failure, vasculitis and glomerulonephritis. In addition, more than 50% of patients with SLE have anemia. One treatment is RBC transfusion, which is the most common procedure performed in U.S. hospitals. RBCs are matched for the ABO and RhD antigens of the recipient. However, they are not routinely matched for as many as 500 RBC-expressed minor alloantigens. This exposure leads to production of RBC alloantibodies in more than 20% of patients with SLE. One pathway that promotes autoantibody production and disease severity in SLE is type 1 interferon (IFNα/β) activation. We found that IFNα/β also critically regulates RBC alloimmunization following transfusion of murine models, including an IFNα/β-dependent model of lupus. Current studies utilize the Cedars-Sinai Lupus Clinical Repository to examine RBC alloimmunization in an IFNα/β-independent mouse model and in patients with SLE.
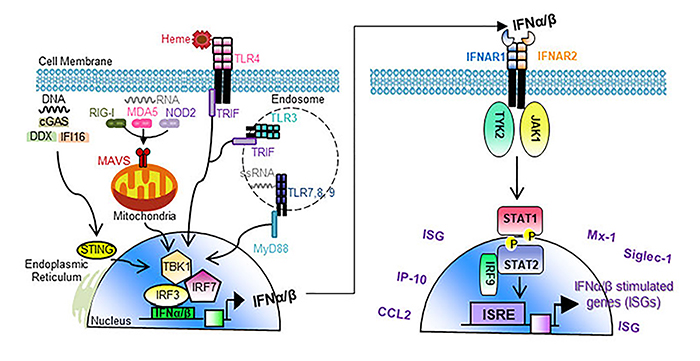
IFNα/β-inducing pathways and signaling. Left: IFNα/β-inducing ligands (nucleic acids and heme), pattern recognition receptors (PRRs), and signaling pathways. Distinct pathway mediators (STING, MAVS, TRIF, MyD88) lead to TBK1 activation of IFNα/β-inducing transcription factors (IRF3 and 7). Right: IFNα/β receptor (IFNAR1/2) signaling results in the production of IFNα/β stimulated genes (ISGs) that are critical for anti-viral immunity and promote RBC alloantibody responses.
Production and consequences of type 1 interferons in patients with sickle cell disease (SCD)
Patients with SCD have the highest frequency of RBC alloimmunization (30%-50%). Studies from our lab and others recently reported that patients with SCD have elevated IFNα/β activity. Current experiments aim to identify the mechanisms, including the triggers and receptors, that lead to IFNα/β production in SCD and to elucidate the consequences (including RBC alloimmunization) of IFNα/β activity in patients with SCD. These projects utilize a murine model of SCD and a cohort of patients with SCD who are seen by hematology collaborators at Cedars-Sinai and USC.