Research Areas
Understanding the Interferon Signaling Pathway in Lupus Neutrophils
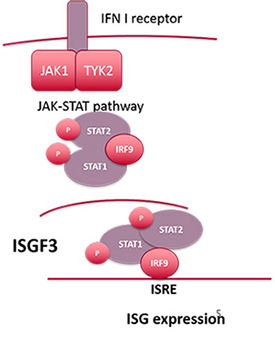
The interferon (IFN) pathway is a key drug target in systemic lupus erythematosus (SLE); therefore, understanding how this pathway is regulated in the context of different immune settings is essential for directing new therapeutic efforts. Lupus neutrophils are highly inflammatory and have been implicated by a number of groups in playing a role in organ pathogenesis. Work in the Jefferies Laboratory has shown that neutrophils from patients with high disease activity have a strong interferon gene signature compared with those of patients with quiescent disease. The Jefferies Laboratory is currently investigating this at the molecular level with the aim of characterizing in detail IFN-driven pathways in these cells and assessing how RNA/DNA sensing pathways affect IFN-driven responses in SLE neutrophils.
Investigating Crosstalk Among Estrogen, RNA/DNA Sensing and IFN in the Immune Response in SLE
In recent years, the Jefferies Laboratory has identified a number of estrogen-regulated cytokines such as interleukin-23 and noncoding RNAs that may be important in driving monocyte and neutrophil activity in lupus, providing important insights into disease pathogenesis and disease susceptibility. Specifically, recent work has shown that mir-302d targets a key component of the transcriptional complex — the transcription factor interferon regulatory factor 9 (IRF9) — that drives the interferon gene signature associated with high disease activity in SLE. Work in the Jefferies Lab has shown that mir-302d is decreased in SLE patient monocytes and neutrophils, resulting in enhanced levels of IRF9, which drives enhanced expression of IFN-dependent genes. This work has important implications for understanding the mechanisms regulating IFN-stimulated genes and suggests that some effects may be independent of the IFN receptor.
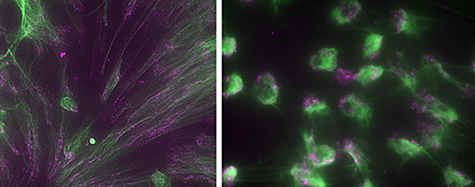
Lupus neutrophils stimulated with PMA, stained with sytox green to detect extruded DNA and anti-MPO (purple) to detect myeloperoxidase associated with NETS
Neutrophils, Endoplasmic Reticulum Stress and Lung Disease
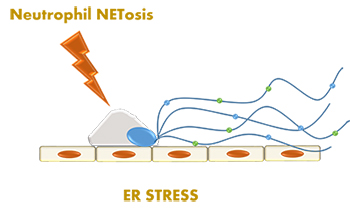
Severe lung disease in lupus is a rare but devastating phenomenon with a survival rate below 50 percent. Patients who do survive have severely reduced lung function as a result. Indeed, it is suspected that over 50 percent of lupus patients have some sort of lung involvement and inflammation, resulting in loss of function and breathlessness, reducing their quality of life significantly. To date there is little known about the pathophysiology of lung involvement in lupus, although hints from analysis of rare genetic autoimmune disease with similarities to lupus are starting to become uncovered. Studies in the Jefferies Laboratory using a mouse model of lupus lung disease have uncovered a role for neutrophils in driving lung inflammation in lupus. Research results point to neutrophil NETosis as the driver of a form of cell death called endoplasmic reticulum stress in lung epithelium, potentially the ultimate driver of loss of lung function observed in patients. The Jefferies Laboratory aims to investigate in detail the role of these neutrophils in lupus lung disease, specifically with respect to their effects on lung epithelial cells and NETosis.
Contact the Jefferies Lab
8700 Beverly Blvd.
Davis Building, Room 5094E
Los Angeles, CA 90048