Research Areas
The Riera Laboratory studies the role of central and peripheral sensory neural circuits on metabolic health and aging by using leading-edge mouse genetics, novel molecular technologies applied to neuroscience and molecular physiology. The missions of the Riera Lab are to discover novel target genes playing a fundamental role in the onset of diabetes and obesity and design preclinical strategies to ameliorate metabolic function in mouse models of disease.
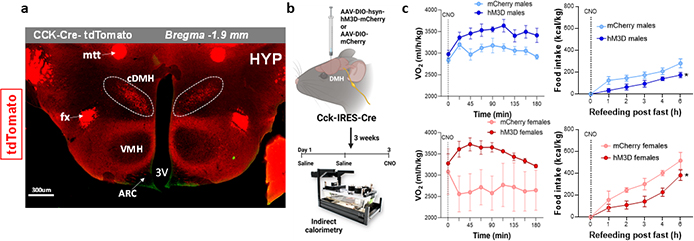
Dorsomedial hypothalamus in the regulation of energy balance. A. tdTomato expression the hypothalamus (HYP) in CCK-cre; Ai14 female mice showing select presence of CCK in neurons from the compact DMH (cDMH). CCK neuronal projections are visible in the arcuate nucleus (ARC), fornix (fx) and mamillothalamic tract (mtt). B. Experimental design for chemogenetic manipulation of Cck-expressing neurons in the DMH. c. Oxygen consumption (VO2) is robustly increased upon DMH-CCK stimulation; Food intake is reduced following chemogenetic activation of DMH-CCK neurons
Role of Afferent Sensory Fibers in Metabolic Health
Previously, the Riera Laboratory reported the role of the transient receptor potential vanilloid 1 (TRPV1), a chemosensory receptor present on afferent Aδ and C sensory fibers projecting to dorsal root ganglia (DRG), in the onset of metabolic decline with age (Riera et al. Cell. 2014). Lack of TRPV1 neurons decreases circulating levels of calcitonin gene-related peptide (CGRP), a neuropeptide implicated in pain responses, neuro-immune communication and vasodilation. Because CGRP levels increase in old animals, Riera Lab research showed that high CGRP levels cause metabolic decline in wild-type animals through blockade of insulin secretion with age. Importantly, pharmacological inhibition of CGRP receptors restores metabolic health in old mice and improves age-dependent loss in insulin secretion. These data highlight a role for the neuropeptide CGRP as a critical regulator of metabolic flexibility upon aging and suggest that DRG fibers may contribute to metabolic homeostasis through neuropeptidic communication with visceral organs. Research in the lab will aim at characterizing the role of CGRP and sensory fibers in visceral tissues to modulate physiological functions and impact on metabolic regulation.
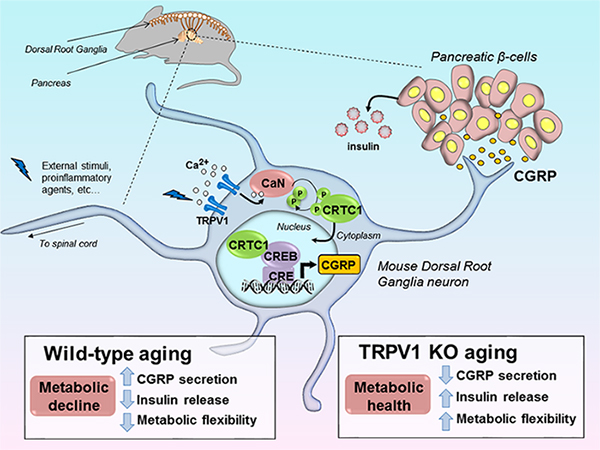
Model for the neuroendocrine regulation of metabolism by transient receptor potential vanilloid subfamily, member 1 expressing neurons. Riera et al. Cell. 2014;157(5):1023-1036. http://www.cell.com/cell/fulltext/S0092-8674(14)00481-4.
Evaluation of Central Olfactory Circuits of Metabolic Control
Obesity is often associated with abnormal feeding behavior implicating central nervous system motivation and feeding centers. The sense of smell is intrinsically connected to food palatability; however, whether olfactory inputs affect energy homeostasis remained unclear. The Riera Laboratory investigates how olfactory sensory neurons (OSNs) regulate whole body metabolism and energy homeostasis. Recently, the lab demonstrated that acute loss of smell perception after obesity onset not only abrogates further weight gain but also improves fat mass and reduces insulin resistance (Riera et al. Cell Metabolism. 2017). Reduced olfactory input activates sympathetic nerve activity, resulting in stimulation of β-adrenergic receptors (β-AR) on white and brown adipocytes to promote lipolysis. Conversely, conditional ablation of the insulin-like growth factor 1 receptor in OSNs enhances olfactory performance in mice by improving OSN neurogenesis and leads to increased adiposity and insulin resistance. These findings unravel a new bidirectional function for the olfactory system in controlling energy homeostasis in response to sensory and hormonal signals.
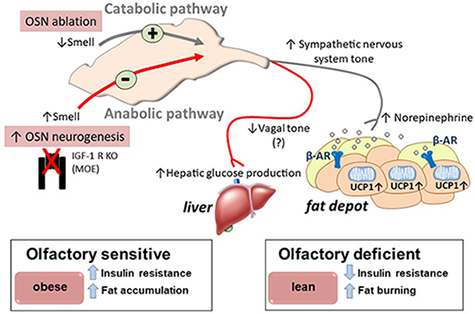
Model for olfactory control of energy balance. Riera et al. Cell Metab. 2017;26(1):198-211.e5. http://www.cell.com/cell-metabolism/fulltext/S1550-4131(17)30357-1.
Contact the Riera Lab
127 S. San Vicente Blvd.
Advanced Health Sciences Pavilion, Suite A8305
Los Angeles, CA 90048