Research Areas
Current research in the Slomka Laboratory focuses on developing innovative methods for fully automated analysis of nuclear cardiology data using novel algorithms and machine learning techniques, and on the development of integrated motion-corrected analysis of positron emission tomography (PET)/computed tomography (CT) angiography imaging.
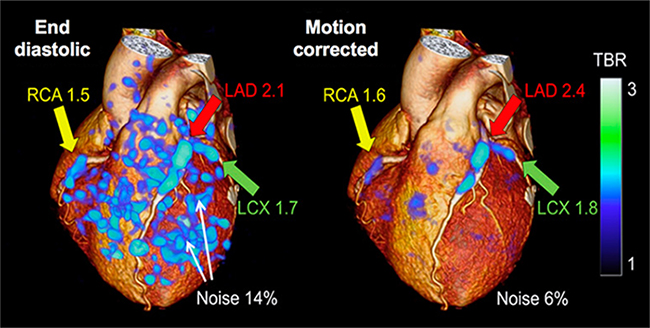
This research was originally published in the Journal of Nuclear Medicine. Rubeaux M, Joshi NV, Dweck MR, Fletcher A, Motwani M, Thomson LE, Dey D, Li D, Berman DS, Newby DE, Slomka PJ. Motion correction of 18F-NaF PET for imaging coronary atherosclerotic plaques. J Nucl Med. 2016;57(1):54-59. http://jnm.snmjournals.org/content/57/1/54.full. © by the Society of Nuclear Medicine and Molecular Imaging, Inc.
The figure above shows noise decrease and target-to-background ratio improvement in 18F-sodium fluoride PET images displaying a 3-D rendering of end-diastolic image (left) and a motion-corrected image (right) superimposed on CT angiography 3-D rendering. Increased uptake is seen in all coronary arteries but it is difficult to differentiate from the noise in the end-diastolic image and becomes clearly visible in the motion-corrected image.
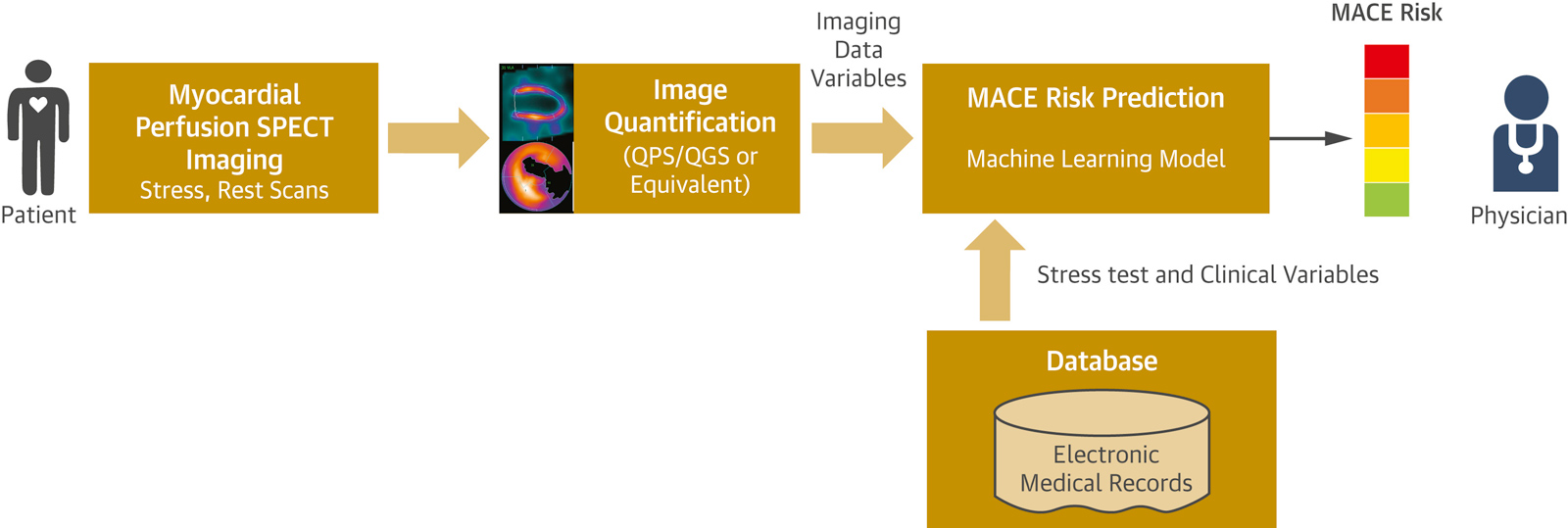
With permission from Betancur J, Commandeur F, Motlagh M, et al, Prognostic Value of Combined Clinical and Myocardial Perfusion Imaging Data Using Machine Learning, JACC Cardiovasc Imaging, 11 (17), 1000-1009, Elsevier, 2018.
The figure above illustrates how our proposed machine-learning model would allow prediction of the risk of major adverse cardiac events for a patient by automatically integrating the clinical data with the imaging data.
Quantitative Prediction of Disease and Outcomes from Next Generation SPECT and CT
Coronary artery disease remains a major public health problem worldwide. It causes approximately one of every six deaths in the U.S. Imaging of myocardial perfusion (delivery of blood to the heart muscle) by myocardial perfusion single photon emission tomography (MPS) allows physicians to detect disease before heart attacks occur, and is currently used to predict risk in millions of patients annually.
The Slomka Laboratory has established an international multisite registry (REFINE SPECT) with all imaging data, diagnostic correlations and prognostic outcomes of more than 23,000 scans. Using this registry, the Slomka Lab has demonstrated that a combination of MPS image analysis and artificial intelligence (AI) tools achieved superior predictive performance compared to visual assessment by experienced readers or current state-of-the-art quantitative techniques.
The overall objective of this research is to optimize the clinical capabilities of MPS in risk prediction and treatment guidance by integrating all available imaging and clinical data with state-of-the-art AI methods.
Specifically, the Slomka Laboratory aims to
- Expand and enhance REFINE SPECT including CT and MPS flow data
- Develop fully automated techniques for all MPS and CT image analysis
- Apply explainable deep-learning, time-to-event AI models for optimal prediction of MACE and benefit from revascularization from all image and clinical data.
Research in the Slomka Lab will result in an immediately deployable clinical tool, which will optimally predict risk of adverse events and establish the relative benefits from specific therapies, beyond what is possible by subjective visual analysis and mental integration of all imaging (MPS, CT, flow), and clinical data by physicians. Most importantly, this research will allow patients to benefit from increased precision and accuracy in risk assessment, thereby optimizing the use of imaging in guiding patient management decisions and ultimately improving outcomes.
Integrated Analysis of Coronary Anatomy and Biology using 18F-Fluoride PET and CT Angiography
Each year, 735,000 Americans have an acute myocardial infarction (heart attack), and approximately 120,000 die from it. Heart attacks occur most commonly due to rupture of atherosclerotic plaques in coronary arteries. Despite this, current diagnostic and treatment algorithms make no allowance for the assessment of disease activity, and currently, all patients with atherosclerosis are treated in a similar manner. This failure to differentiate stable from an active disease may result in potentially unnecessary or insufficient therapies.
In a breakthrough series of studies, Slomka Laboratory collaborators at the University of Edinburgh discovered that positron emission tomography (PET) with 18F-sodium fluoride (18F-NaF; an inexpensive and widely available tracer approved by the Food and Drug Administration) can readily identify plaque rupture and increased coronary plaque activity. The Slomka Lab proposes to build further on this success by addressing several important remaining limitations that prevent us from translating this technology to broad clinical use. The limitations include complicated and subjective image analysis, underutilization of the concomitant coronary computed tomography angiography (CTA) for plaque characterization, inability to utilize prior CTA for the analysis of 18F-NaF PET, lack of methods to integrate all available PET and CTA data, and significant motion of the coronaries during the PET scan.
The Slomka Laboratory will use a multifaceted approach to automate and improve coronary 18F-NaF PET imaging by full integration with CTA and correction for cardiac, respiratory and patient motion. The overall goal of the proposal is to optimize the measurement of disease activity in coronary atherosclerosis using integrated 18F-NaF PET/CTA imaging, with the opportunity to validate this development against clinical outcomes in a real-world, multicenter patient study.
In this work, the Slomka Lab aims to
- Integrate quantification of CTA and PET image data
- Develop new methods for simultaneous correction of cardiac, respiratory and patient motion for coronary PET
- Clinically evaluate new methods in a multicenter clinical trial (separately funded and already underway), further refining risk prediction for heart attacks with integrated PET+CTA risk score derived by machine learning
This work will lead to a robust and reproducible clinical method for stratification of patients for risk of heart attacks, with potential for application in identifying patients who would most benefit from expensive and potentially risky treatments. Our techniques could also be used in future clinical trials to test the efficacy of novel therapies. Moreover, the new analysis will be applicable to other PET tracers that may be developed to investigate other pathological processes in the coronary vasculature. The resulting software will be shared with clinical institutions performing coronary PET to facilitate standardization and automation of this novel plaque imaging technique.
Contact the Slomka Lab
8700 Beverly Blvd.
Suite Metro 203
Los Angeles, CA 90048