Research Areas
In order to study molecular mechanisms of inflammation and their contribution to inflammatory disease, the Stehlik and Dorfleutner Lab routinely employs molecular, cellular, biochemical and imaging approaches in human and mouse macrophages, combined with in vivo mouse inflammatory disease models. The lab is further evaluating our findings in human samples, including tissue and serum from inflammatory disease patients to assess their relevance for human disease.
Inflammasomes
Inflammasomes are protein complexes that link recognition of danger signals to the activation of inflammatory caspases, including caspase-1, -4, -5, -8 and -11, through canonical, non-canonical and alternative pathways. Consequentially, caspase activation leads to the proteolytic maturation and secretion of the pro-inflammatory cytokine substrates IL-1β and IL-18, to inflammatory cell death called pyroptosis, and to the release of danger signals (DAMPs). Excessive release of IL-1β and IL-18 in particular has been directly linked to the pathology of a wide spectrum of immune diseases. Hence, for developing novel therapies, it is crucial to better understand inflammasome biology, including the specific function and activation as well as inhibition mechanisms of cytosolic sensors, including the NOD-like receptors (NLRs) and the AIM2-like receptors.
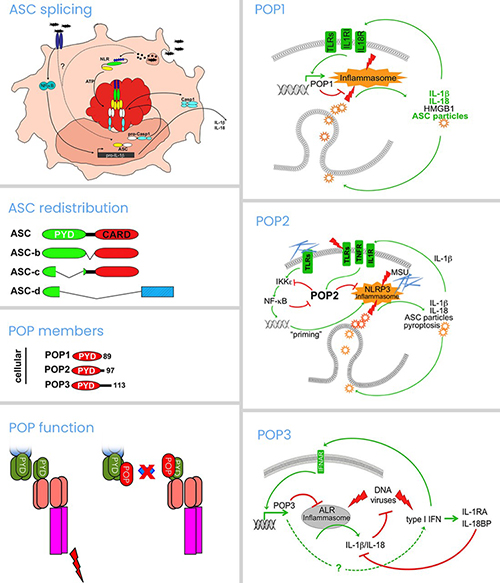
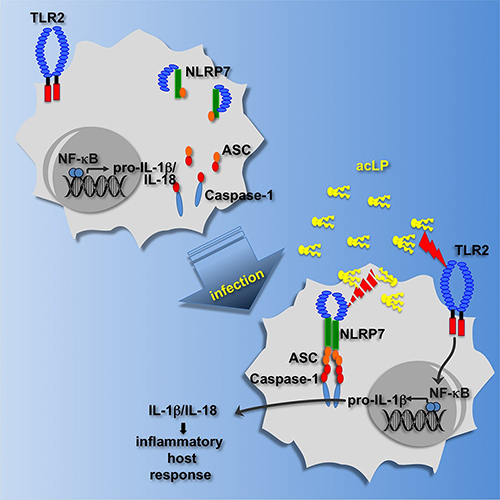
Cellular mechanisms include the disruption of the inflammasome complex by PYD-only proteins (POPs) and CARD-only proteins (COPs), as well as post-translational protein modifications. Our research contributed to the discovery of several key aspects of inflammasome biology and the inflammatory response. The Stehlik and Dorfleutner Lab generated several unique mouse models for studying inflammasome regulation in vivo. We discovered ASC as the essential adaptor for caspase-1 activation, were the first to visualize the endogenous inflammasome speck in macrophages and described several mechanisms by which ASC regulates inflammasomes, including by induced subcellular redistribution and splicing. The lab also discovered the NLRP7 inflammasome in human macrophages. In addition, we described the function of COPs and discovered the first, and subsequently all other POPs, thereby establishing the POP family of inflammasome regulators. We described their unique role in vivo and generated an inflammasome-targeting proof-of-concept therapy for inflammatory disease. Current work focuses on several novel- inflammasome components that either positively or negatively regulate the response and how these proteins shape inflammatory and autoimmune disease.
Inflammasome Regulation
Inflammasome assembly is mediated by nucleation of the adaptor ASC by upstream pattern recognition receptors and this step requires interaction via specific death domain fold-containing protein-protein interactions, namely the PYRIN domain (PYD) and caspase recruitment domain (CARD). The Stehlik and Dorfleutner Lab observed significant species differences in the repertoire of these proteins in humans and mice and discovered all members of a family of small, single PYD-containing PYRIN domain-only proteins (POPs) and one of the CARD-only proteins (COPs), which are encoded in humans but are lacking in mice. Our work demonstrated a key role of these proteins in shaping the immune response and their ability to ameliorate inflammatory disease. Work in humanized transgenic mice with cell type-specific or inducible expression, spontaneous and induced inflammatory and immune disease models, and in human cells and tissues is ongoing, to further elucidate their role in innate immunity.
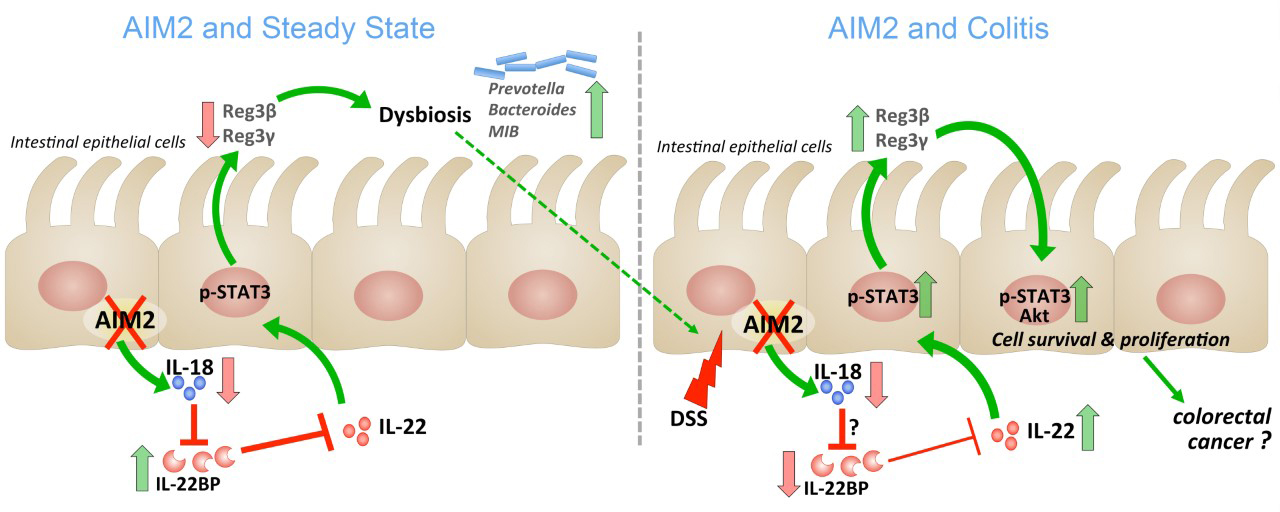
Maintenance of Intestinal Homeostasis
Tolerance toward commensal bacteria is key for promoting microbial diversity, maintaining intestinal homeostasis, preventing pathogen infection and systemic disease by shaping immune responses also outside of the intestine. Our ongoing studies aim to elucidate the molecular mechanisms involved in the maintenance of this tolerance and investigate events that lead to a break in tolerance for instance as in inflammatory bowel disease, colorectal cancer and other diseases.
Molecular Defects of Colorectal Cancer, Choriocarcinoma and Breast Cancer
The Stehlik and Dorfleutner Lab investigates to delineate molecular defects contributing to the development of select carcinomas in the colon, uterine and breast, focusing on cytosolic DNA sensing, defects in the NLRP7 inflammasome—discovered by us—and AFAP1, respectively, utilizing novel mouse models and studies in human tissues
Contact the Stehlik & Dorfleutner Lab
127 S. San Vicente Blvd.
Pavilion, Room A-8700
Los Angeles, CA 90048