Research Areas
Overarching interests of the laboratory are to define epithelial stem and progenitor cells that maintain the human lung, to define mechanisms that regulate quiescence versus activation of these reparative cells and renewal versus differentiation, in the setting of normal tissue maintenance and lung disease. Our studies benefit tremendously from the participation of patients receiving clinical care for lung disease, who generously provide access to clinical samples that are used to identify candidate cellular and molecular targets for initiation and progression of chronic lung disease. Three-dimensional culture systems and genetically defined animal models are used to define mechanisms of disease and validate therapeutic targets. Areas of research focus include:
Structural Remodeling of the Lung in Chronic Lung Disease
Lung tissue remodeling, suboptimal repair leading to loss of structure and function, is pathognomonic of most chronic lung diseases and accounts for progressive declines in lung function, morbidity and mortality. Chronic diseases of small airways and the gas-exchange region have an enormous societal impact both within the U.S. and worldwide. Of the estimated 225,000 deaths attributed to lung disease in the U.S. in 2007, greater than 50% were due to chronic obstructive pulmonary disease (COPD). Less prevalent but with more limited treatment options is idiopathic pulmonary fibrosis (IPF), the most common type of interstitial pneumonia, which has a mean survival time of three years after initial diagnosis and afflicts upward of 0.2% of the population in North America and Europe.
Our studies seek to define cellular and molecular defects within epithelial cells that lead to repair defects and scar formation, and modeling of these events using 3D culture systems and through genetic manipulation in mice. Data below highlight advanced genomics approaches that are being used to define pathways that are defective in epithelial stem cells of the IPF lung and mouse models that are being employed to investigate mechanisms of disease.
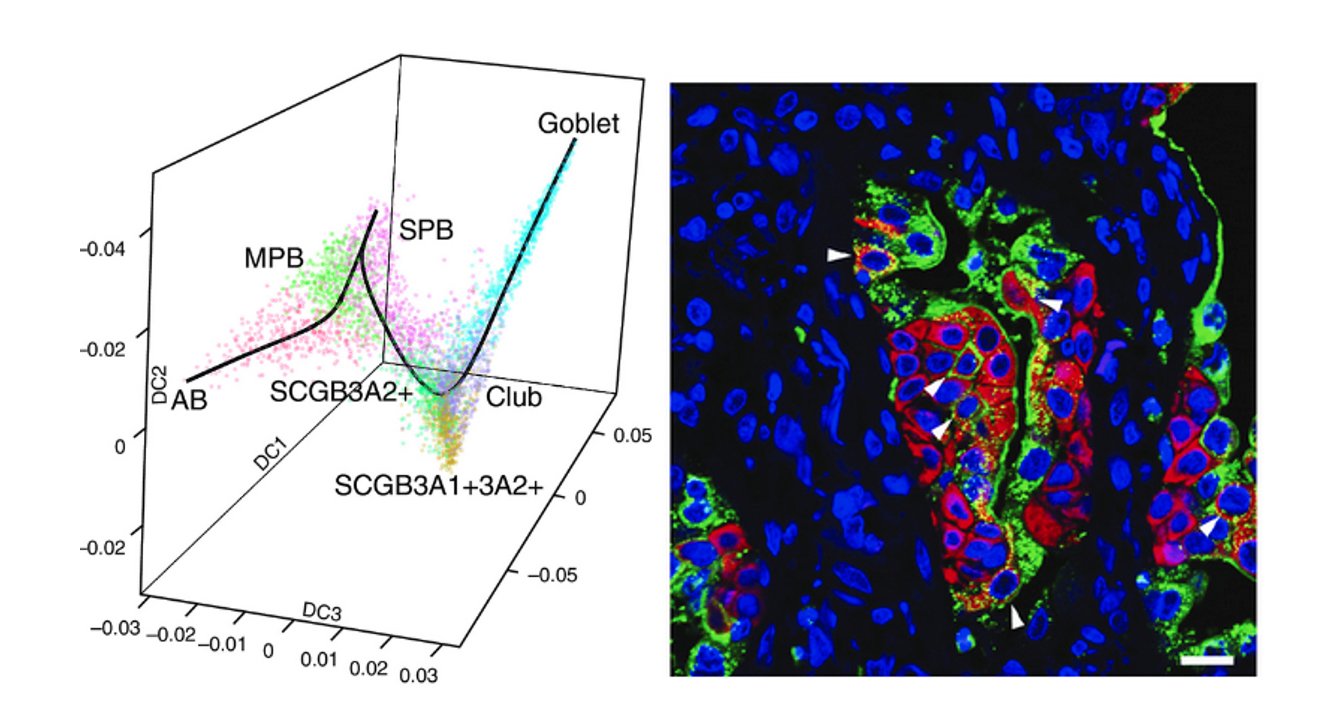
Single cell RNA-Seq reveals the unique molecular phenotype of epithelial cells from lung tissue of IPF patients. Single cell RNA-Seq of epithelial cells isolated from lung explant tissue recovered from patients undergoing transplant for end-stage IPF. Data are visualized by Diffusion Map, colored circles representing the aggregate transcriptome of individual cells with molecular similarity indicated by relative position in three dimensions; colors represent cell types as indicated. These data highlight the unique molecular phenotype of basal cells recovered from IPF explant tissue. Secretory Primed Basal (SPB) cells have a molecular signature that link them directly to secretory cells expressing SCGB3A2. On the right immunoreactivity for KRT5 (red) and CD66 (green) show expansion SPB cells in IPF.
Studies shown below using mouse models show that p53 regulates stem cell quiescence versus activation that may underlie progenitor cell defects observed in fibrotic lung disease, and that a genetic model leading to activation of p53 signaling leads to progressive pulmonary fibrosis.
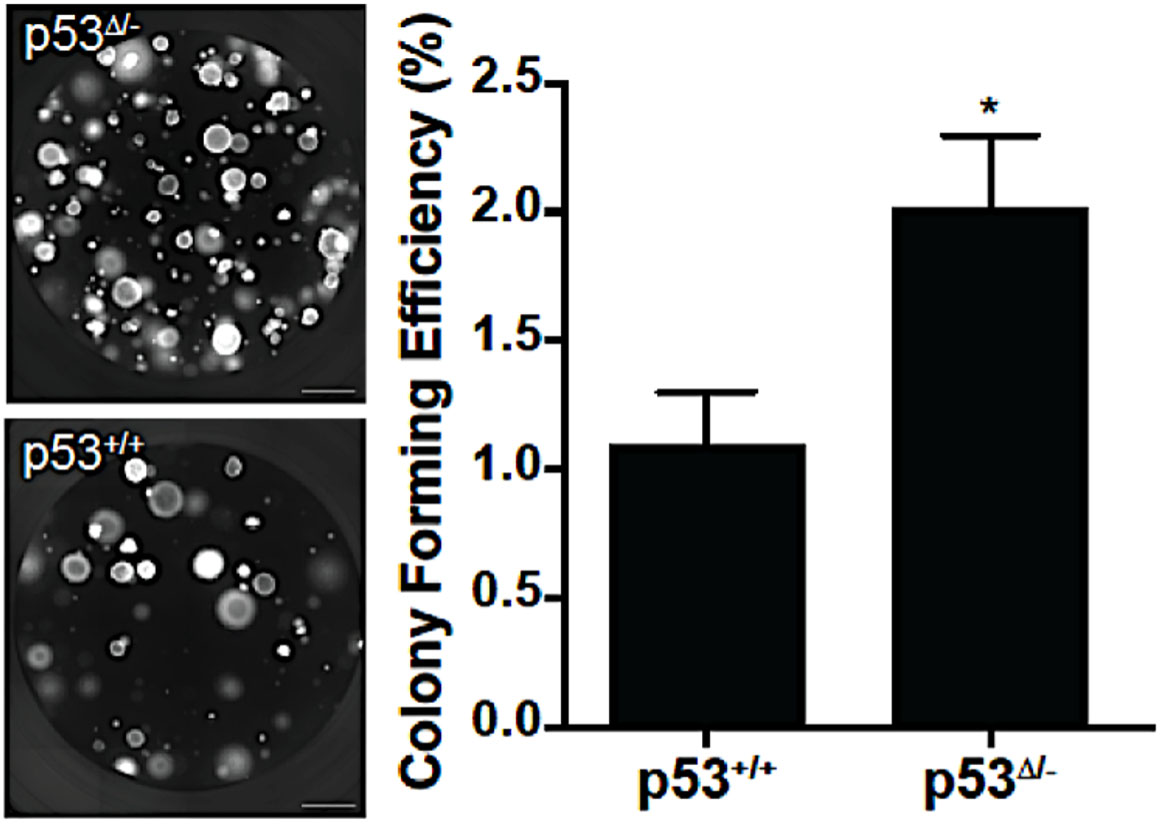
p53 loss-of-function in airway progenitor cells increases colony-forming efficiency (CFE). Scgb1a1 lineage-labeled club cells that were either p53 sufficient (p53+/+) or p53 LOF (p53D/-), were isolated by FACS and placed in 3D organoid cultures. Representative images of seven-day cultures are shown on the left (grayscale), with graphs of quantitative data shown on the right. p53 LOF resulted in a twofold increase in CFE but no change in colony size. For more details see McConnell, et al. (Cell Reports. 17:2173-2182. 2017.)
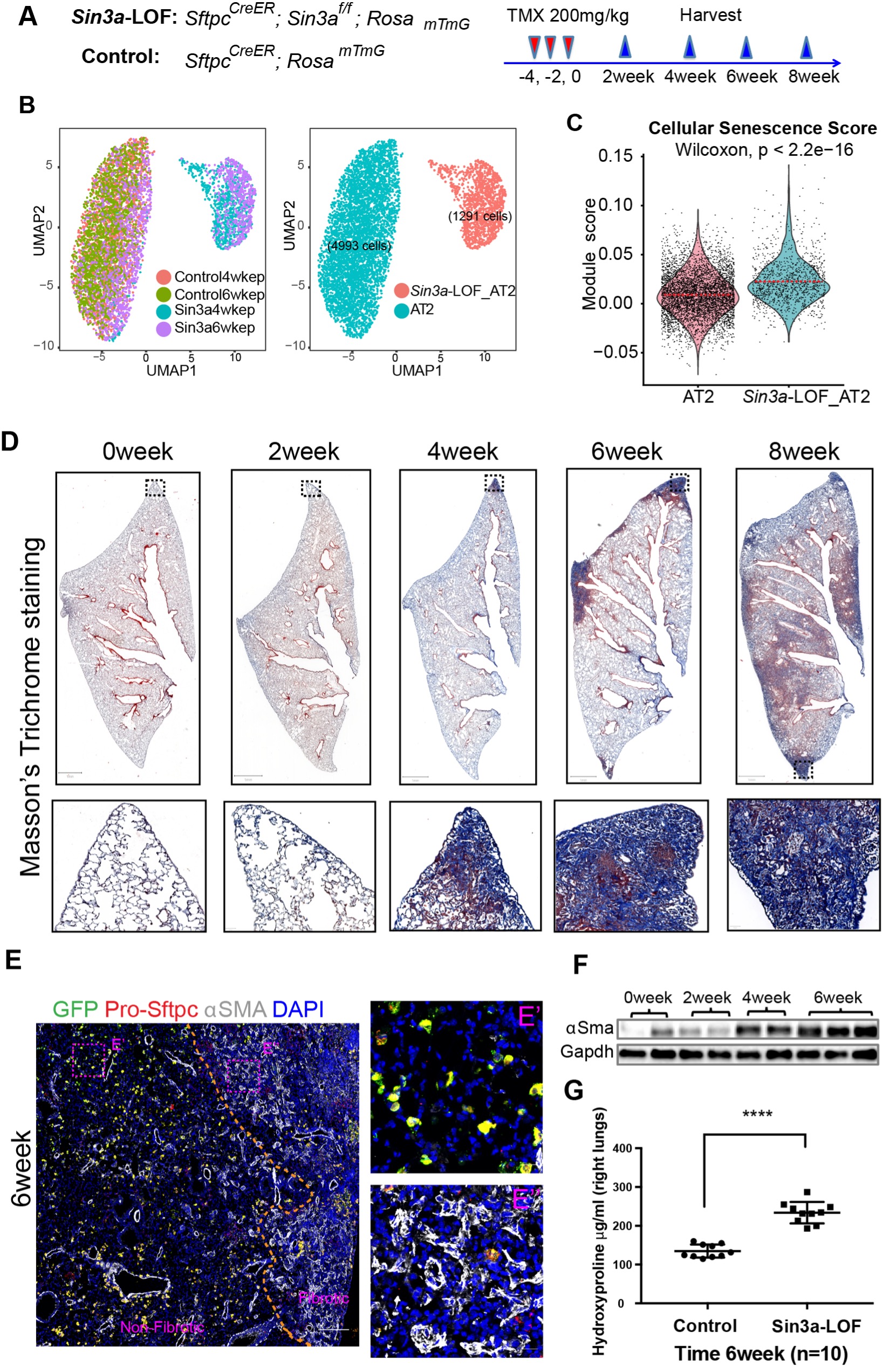
Progressive pulmonary fibrosis following conditional modulation of the epigenome in alveolar type 2 cells.
A) Schematic of experiment design. B) UMAP visualization of AT2 cell subset clustering from mouse epithelial cell scRNA-seq. C) Cellular senescence score comparing Sin3a-LOF AT2 cells with control AT2 cells. D) Masson’s trichrome stain of tamoxifen-exposed Sin3a-LOF mice lung tissue sections shows progressive fibrosis and matrix deposition. E) Representative immunofluorescence staining of Sin3a-LOF lung tissue six weeks after tamoxifen exposure for lineage reporter-GFP (green fluorescent protein), pro-Sftpc and aSMA. The dashed line represents the boundary between fibrotic and nonfibrotic tissue. F) Western blot detection of aSMA abundance within lung tissue of Sin3a-LOF mice. E and F indicates fibroproliferation of Sin3a-LOF mouse lungs. G) Hydroxyproline content of right lung between Sin3a-LOF and control mice six weeks after tamoxifen exposure (n=10 for each group) indicates increased matrix deposition. (AJRCCM. doi: 10.1164/rccm.202004-1274OC)
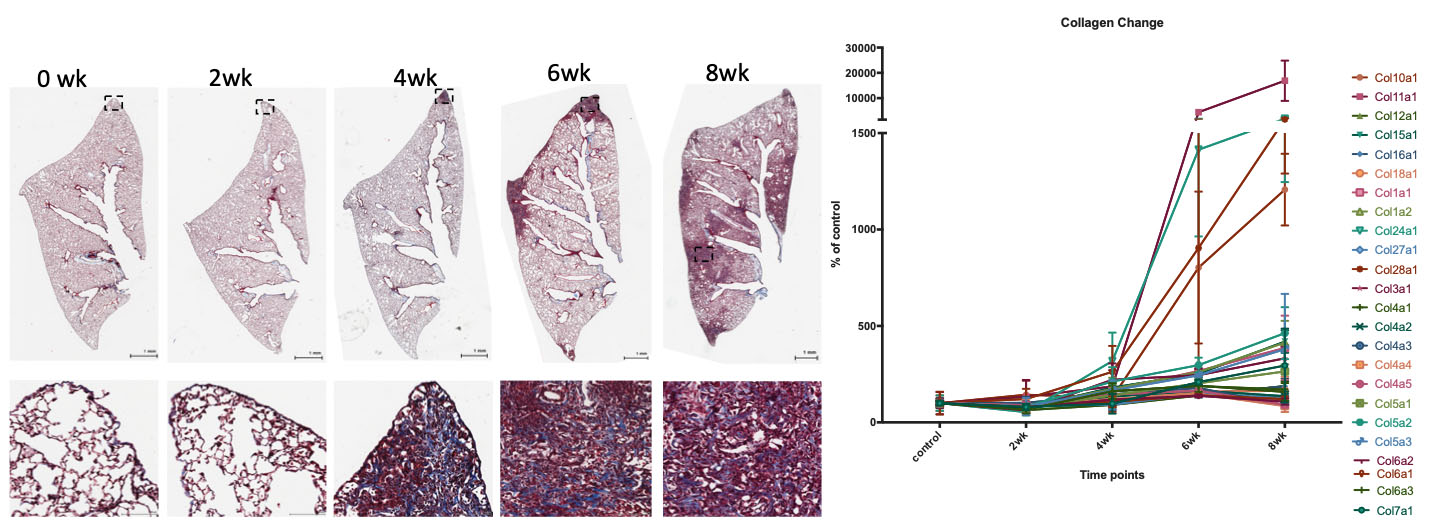
Progressive pulmonary fibrosis following conditional modulation of the epigenome in alveolar type 2 cells. Masson’s Trichrome stain of lung tissue sections shows progressive fibrosis and matrix deposition with time following introduction of a genetic lesion that alters histone deacetylase function in alveolar type 2 cells (left panels). RNA-Seq analysis of total RNA recovered from lungs of mice show time-dependent increases in collagen gene expression reflective of increased matrix production and fibrosis.
Our studies seek to establish preclinical mouse models of COPD to understand basic mechanisms that drive initiation and progression of disease and for validation of candidate therapies.
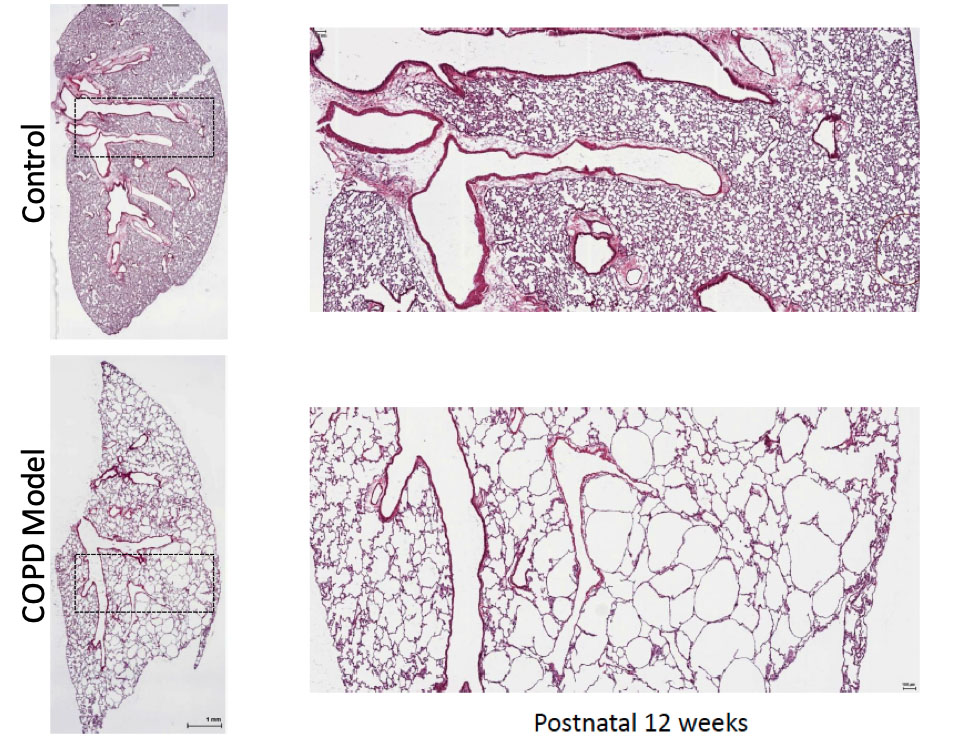
Progressive postnatal emphysema in mice following gene alterations regulating histone modifications in lung mesenchyme. H&E stained lung tissue from representative 12-week-old female mice that are either wild type (control) or functionally defective in their ability to post-translationally modify histone proteins in lung mesenchyme. Mice have normal lungs at birth but develop progressive emphysema postnatally. This model is being used to determine roles for lung mesoderm in postnatal tissue maintenance and as a preclinical model to test novel therapies to repair emphysematous lung tissue remodeling.
Monogenic Diseases
Cystic fibrosis is a monogenic disorder affecting approximately 1 in 2500 births or an estimated 70,000 individuals worldwide. The underlying genetic defect involves mutations that impact functionality of the cystic fibrosis transmembrane conductance regulator (CFTR), leading to defects in electrolyte transport. The most pronounced clinical manifestations of cystic fibrosis result either from epithelial dysfunction and mucus plugging in many different tissues including the upper and lower respiratory tract, gastrointestinal tract and reproductive tract, or from defects in nutrient and salt absorption/secretion. The advent of novel pharmacologic approaches to modulate CFTR trafficking or function has provided effective therapeutic options for approximately 75% of patients. However, improved options that go beyond palliative therapy are desperately needed for the thousands of patients that are not responsive to CFTR modulator therapy. Our goals are to provide an improved understanding of epithelial defects observed in lungs of patients with CF lung disease and to develop stem cell replacement therapies aimed at providing treatment options to patients that are not responsive to current drug therapies.
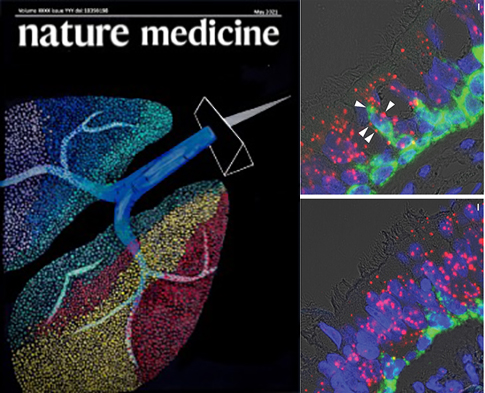
Single cell RNA-Seq reveals the unique molecular phenotype of epithelial cells in lungs of patients with CF lung disease. Cystic fibrosis (CF) is a lethal autosomal recessive disorder that afflicts more than 70,000 people worldwide. People with CF experience multi-organ dysfunction resulting from aberrant electrolyte transport across polarized epithelia due to mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene.
In a recent study (cover of Nature Medicine, on left) we provide new clue into the causes of CF. Image on the right show changes in the composition of cell types lining the airways in CF. Arrows indicate basal cells (green for KRT5) in parabasal position, co-expressing a marker of early ciliogenesis (red for LRRC6).
Lung Cancer
Lung cancer remains the leading cause of cancer-related deaths both nationally and worldwide. Of lung cancer cases, non-small cell lung cancers (NSCLC) are the most prevalent with adenocarcinoma (ADC) and squamous cell carcinoma (SCC) accounting for 85% of all NSCLC cases. Adenocarcinomas include a rage of overlapping sub-histotypes that collectively represent the most common subtype of NSCLC. A pervasive problem in the treatment of all cancers is their ability to undergo somatic mutations leading to the generation of a heterogeneous population of tumor cells, each of which has the potential to show unique profiles of sensitivity to commonly used chemotherapeutic drugs.
Our studies seek to define tumor cell heterogeneity within patient-derived primary lung adenocarcinoma samples through use of state-of-the-art technologies for single cell molecular profiling and to use in vitro 3D cultures for disease modeling.
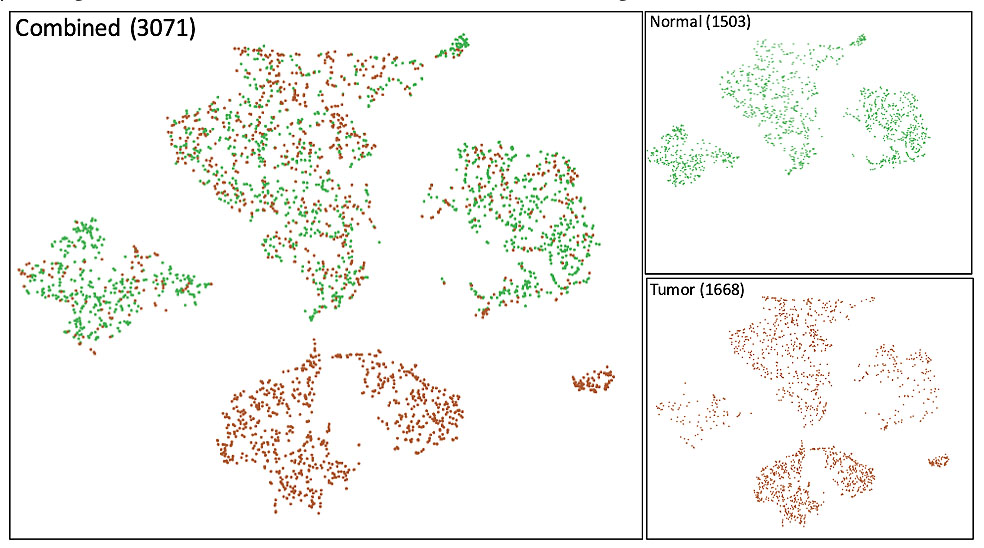
Single cell RNA-Seq reveals tumor cell heterogeneity in stage 3 lung adenocarcinoma. Tumor tissue and surrounding normal tissue was obtained from a nonsmoking patient undergoing lobectomy to treat primary lung cancer. Single cell RNA-Seq was performed to profile the transcriptome of epithelial cells isolated from tumor and non-tumorigenic adjacent tissue. Results are presented as t-distributed stochastic neighbor analysis (tSNE) plots of either non-tumorigenic normal cells (upper right in green) tumor cells (lower right in brown) or merged datasets (left). Tumor cells include a fraction of normal cells that overlap with epithelial cells in non-tumorigenic tissue and a distinct subset of tumor cells. Three major sub-groups of tumor cells are observed reflecting tumor cell heterogeneity.
Acute Lung Injury
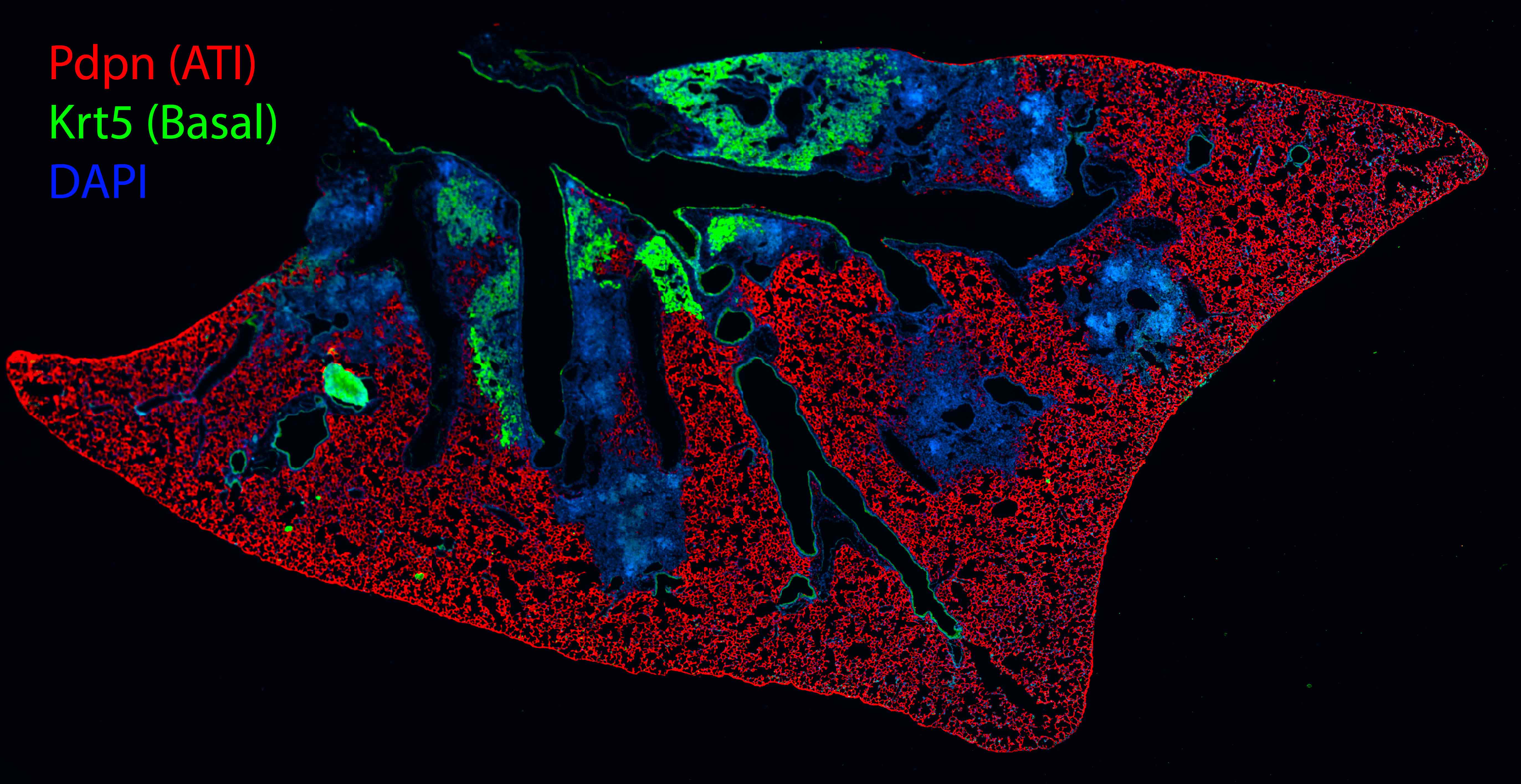
Alveolar destruction and structural lung remodeling following infection of mice with H1N1 influenza virus. Shown is a section of lung tissue prepared from a mouse infected with H1N1 influenza virus and recovered for 14 days. Alveolar type 1 cells are shown by immunostaining for podoplanin (Pdpn; red immunofluorescence) and airway basal cells stained with cytokeratin 5 (Krt5; green immunofluorescence); nuclei are stained with DAPI (blue). Regions of alveolar destruction are shown by loss of Pdpn immunoreactivity. Expanding populations of airway basal cells are seen in green, which colonize damaged alveolar regions leading to aberrant tissue remodeling.
Lung tissue remodeling following acute respiratory viral infection
Basal cell (BC) hyperplasia is a common pathological feature of chronic lung disease and acute H1N1 influenza virus infection. However, the cell types that contribute to their formation and impact of surrounding microenvironmental cues that influence the behavior of BCs have yet to be fully elucidated.
To address this knowledge gap, we have employed single cell transcriptomics and dual recombinase fate-mapping in mice to assess dynamic changes in the molecular landscape and fate of lung epithelium following influenza infection. Our studies define a rare subpopulation of intralobar serous cells as a progenitor cell-of-origin for nascent BC that expand in airways and repopulate the injured gas-exchange region following viral infection. Additionally, we show that the balance between BC renewal versus differentiation into serous-like cells is influenced by innate immune activation and local production of IL-22. In summary, our data show that preexisting intralobar serous cells are a major source of expanding BCs in lungs of influenza virus-infected mice, while their subsequent differentiation back into serous cells is blocked by increased local production of IL-22.
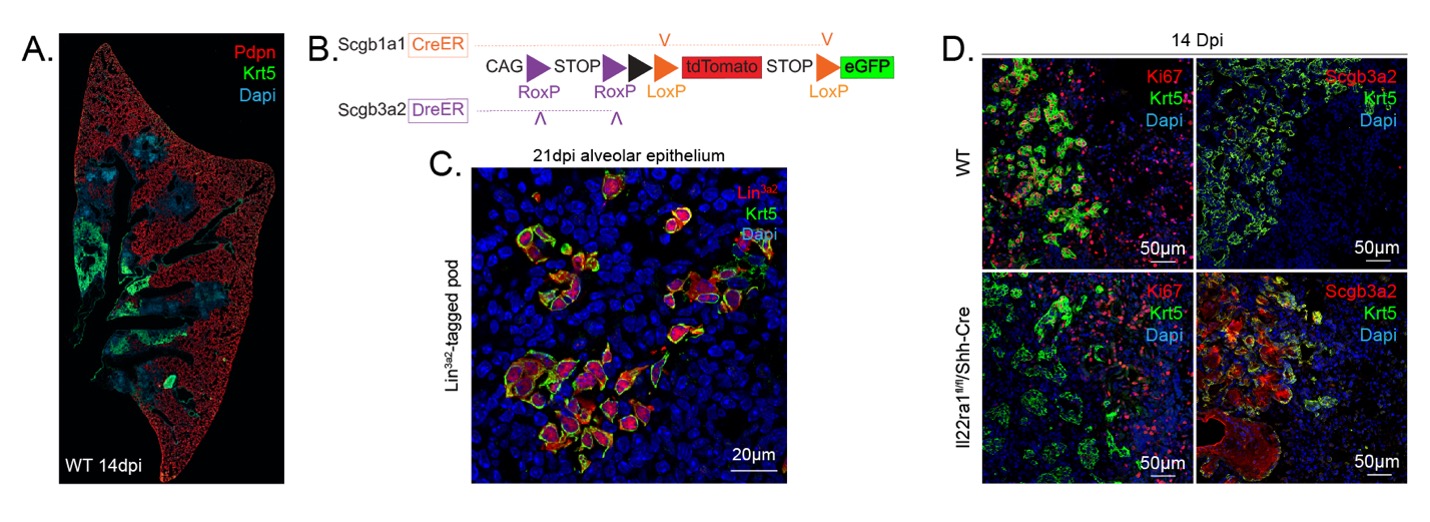
Serous-basal-serous transition and immunomodulation of progenitor cell function by innate immunity following infection of mice with H1N1 influenza virus. A) Shown is a section of lung tissue prepared from a mouse infected with H1N1 influenza virus and recovered for 14 days. Alveolar type 1 cells are shown by immunostaining for Pdpn (red) and airway basal cells stained with Krt5 (green). Regions of alveolar destruction are shown by loss of Pdpn immunoreactivity. Expanding populations of airway basal cells are seen in green, which colonize damaged alveolar regions leading to aberrant tissue remodeling. B) Schematic illustration of recombinase driver and reporter alleles used in Dre/Cre recombinase mice for independent lineage labeling of serous and club cells. C) Representative immunofluorescence colocalization of tdT (Lin3a2; red) with Krt5 (green) among lineage-positive alveolar clusters 21 days after PR8 infection. D) Representative immunofluorescence localization of Ki67, Scgb3a2 (red) and Krt5 (green) in lung tissue of PR8-infected WT and IL-22ra1fl/fl/Shh-Cre mice.
Contact the Stripp Lab
127 S. San Vicente Blvd.
Advanced Health Sciences Pavilion, Room A8600
Los Angeles, CA 90048