Research Areas
Restoring the Normal Sumoylated Proteins Expression in Alcoholic Liver Disease and Cancer Using a Proteomic Approach In Vivo Model
Post-translational modifications play an important role in regulating protein activity by altering their functions. Sumoylation is a highly dynamic process, which is tightly regulated by a fine balance between conjugating and deconjugating enzyme activities. Sumoylation affects intracellular localizations and their interactions with their binding partners, thereby changing gene expression.
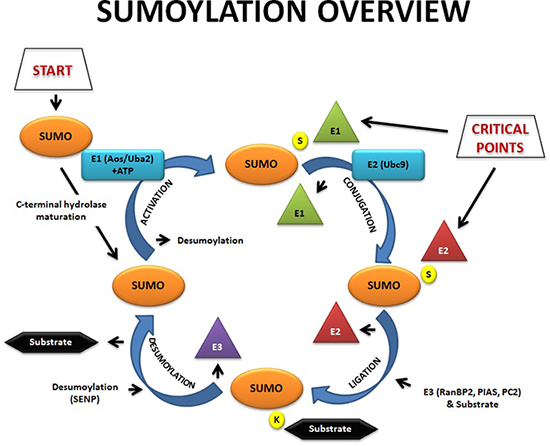
The SUMO modification cycle. Before attachment to proteins, SUMO proteins must be cleaved into their mature forms by SENPs. The processed SUMO proteins are then activated by conjugation to the E1 heterodimer SAE1–SAE2, after which the SUMO is transferred to the E2 enzyme Ubc9. Finally, SUMO is ligated to substrate proteins by an isopeptide bond between the terminal glycine on SUMO and the ɛ-amino group of a lysine in the substrate. The efficiency of the ligation reaction is aided by SUMO ligase E3 proteins, which directly interact with both target proteins and the E2 enzyme, thereby acting as bridging factors to increase the efficiency of this reaction. SUMO polypeptides are removed from target proteins by the action of SENPs, which recovers the SUMO proteins for attachment to other proteins in another cycle of the sumoylation pathway.
These changes in gene expression in turn affect signaling mechanisms that regulate many cellular functions, such as cell growth, proliferation, apoptosis, DNA repair and cell survival. It is becoming apparent that deregulation in the small ubiquitin-related modifier pathway contributes to oncogenic transformation by affecting sumoylation/desumoylation of many oncoproteins and tumor suppressors. Loss of balance between sumoylation and desumoylation has been reported in a number of studies in a variety of disease types, including cancer. The role of sumoylation in alcoholic liver diseases such as steatosis, steatohepatitis, fibrosis, cirrhosis and hepatocellular carcinoma is unknown. Given that alcoholic liver disease (ALD) is the major cause of liver disease in Western countries, the goal of research in the Tomasi Lab is to provide highly novel information on the role of sumoylation in the development of ALD.
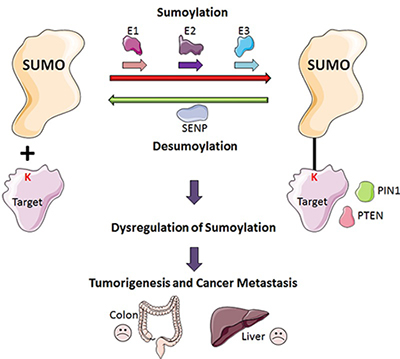
Sumoylation in regulating a wide range of cellular processes. Accumulating lines of evidence support the idea that balanced levels of SUMO-modified and unmodified proteins are required for normal cellular processes, whereas an imbalance in sumoylation often leads to human diseases, including various types of cancer. Many SUMO-specific conjugating and deconjugating enzymes are overexpressed in various human cancers, leading to an imbalance in sumoylation, which is often associated with advanced stages of cancer as well as low survival rates.
Contact the Tomasi Lab
110 N. George Burns Road
Davis Building, Room 3094A
Los Angeles, CA 90048