Research Areas
We perform preclinical, translational and clinical trial research focused on transplantation immunology biology and transplant outcomes. Preclinical and translation work focuses on links between innate and adaptive immunity including complement system. Clinical trials translate the findings to clinical practice using novel therapies and biomarker-guided treatment strategies.
Complement Biology and Transplantation
Preclinical studies in murine models and using human cells demonstrated that the complement system is required for T-cell activation and function through mechanisms involving signaling via C3a receptor and C5a receptor expressed on the surfaces of both antigen presenting cells and T- cells. Separate studies showed that decay-accelerating factor (DAF) regulated complement activation is requisitely involved in the formation and maintenance of germinal center B-cell responses that lead to high-affinity antibody production.
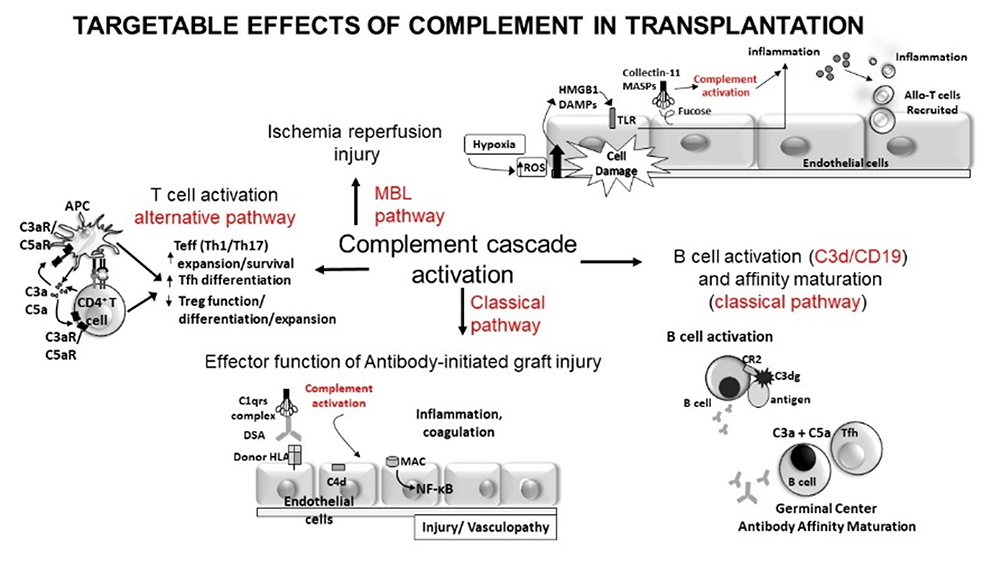
Overview of effects of complement on immune responses relevant to transplantation.
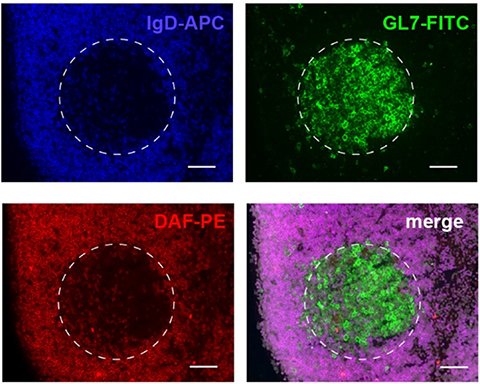
IF staining of germinal center (GC) showing decay accelerating factor (red) is present on the surface of naïve B-cells (blue) but absent on surfaces of GC B-cells (green).
Innate Immune Activation by cGAS-STING and Transplantation
Cyclic GMP-AMP synthase (cGAS) functions as a sensor for intracellular double-stranded DNA and triggers stimulator of interferon genes (STING) pathway to initiate Type 1 interferon responses that amplify inflammation. Increasing evidence indicates that this pathway is an important innate immune trigger of alloreactive T- and B-cells that drive transplant rejection. In collaboration with the Marbán and Ke Labs at Cedars-Sinai (Smidt Heart Institute), we are testing this hypothesis in murine models using novel small non-coding RNAs that inhibit the pathway.
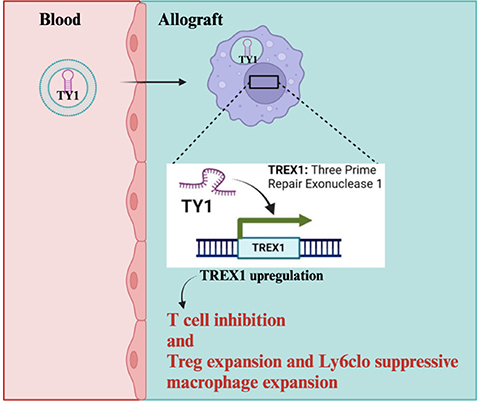
The small non-coding RNA-derived therapeutic TY1 is hypothesized to limit cGAS-STING activation in macrophages through upregulating TREX1, an exonuclease that degrades free intracellular DNA. The effects promote reduced T-cell activation and promote transplant survival.
Butyrophilin2A2 Regulation of Transplant Outcomes
Butyrophilin2A2 is related to costimulatory molecules and is expressed on antigen presenting cells. Collaborative studies published in 2025 with the Karumanchi Lab (nephrology, Cedars-Sinai) showed that Butyrophilin2A2 binds to CD45 on T-cells, limiting activation of naïve and effector T-cells and promoting induction and expansion of regulatory T-cells, together reducing autoimmune kidney disease in animal models. Ongoing work is testing how this molecule impacts alloreactive T-cell and B-cell immune responses that drive transplant rejection using murine models and human cells.
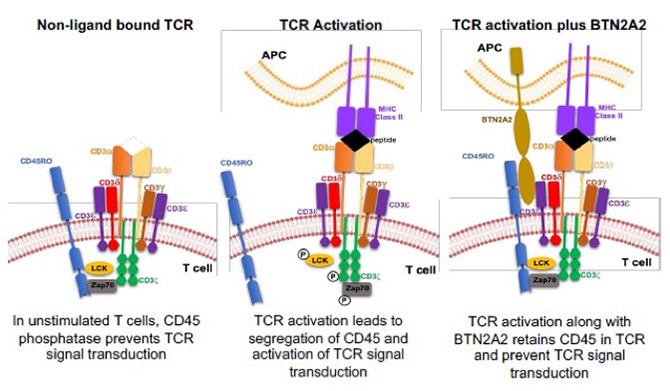
Butyrophilin2A2 alters T-cell activation, limiting effector T-cells and promoting regulatory T-cell induction and expansion.
Clinical Trials
Assessment of Biomarker Guided Calcineurin inhibitor Substitution in Kidney Transplantation (ABCs)
The goal of this study is to prospectively test the strength of the relationship between the HLA molecular mismatch (eplets) and transplant outcomes (rejection, DSA, graft failure), and to test the hypothesis that recipients with a low molecular mismatch to their donor can be safely switched from tacrolimus to the costimulatory blockade inhibitor abatacept, leading to improved kidney function, neurocognitive abilities and patient-reported outcomes. Mechanistic studies will address how the molecular mismatch affects T-cell immunity and the effects of the interventions on graft biology (spatial transcriptomics).
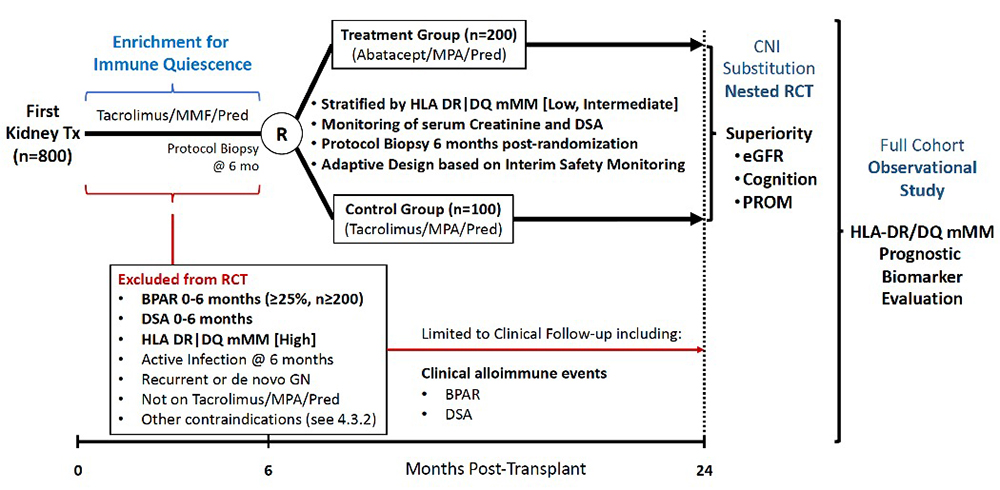
Study design for ABCs trial.
Improving Deceased-Donor Kidney Transplant Outcomes Via a Single Intragraft Injection of C1 Esterase Inhibitor (IMPROVE)
Following deceased donor kidney transplantation, Lectin pathway-initiated complement activation is hypothesized to crucially drive ischemia reperfusion injury that negatively impacts transplant outcomes. Based on results of a single center pilot trial showing that a) C1 esterase inhibitor binds to donor endothelium after an intra-artery injection, and b) the intervention improves kidney graft function—we are performing a large multicenter randomized trial testing the efficacy of this therapy on deceased donor kidney transplant outcomes.
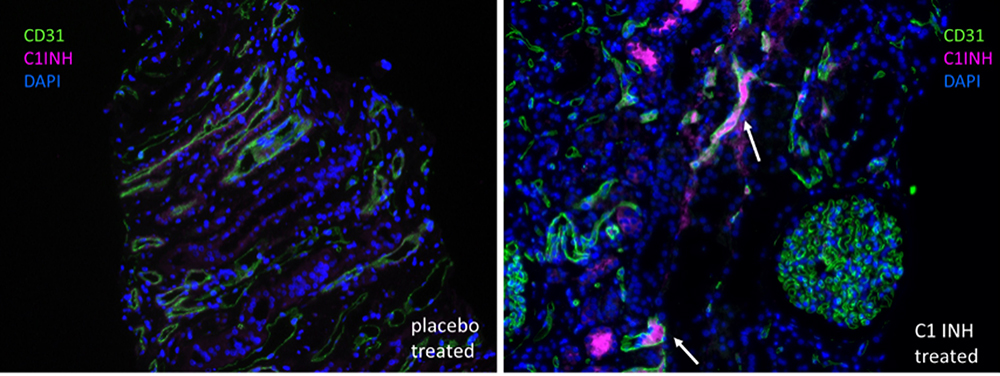
C1 esterase inhibitor is detectable within blood vessels of the donor organ after intra-arterial injection.
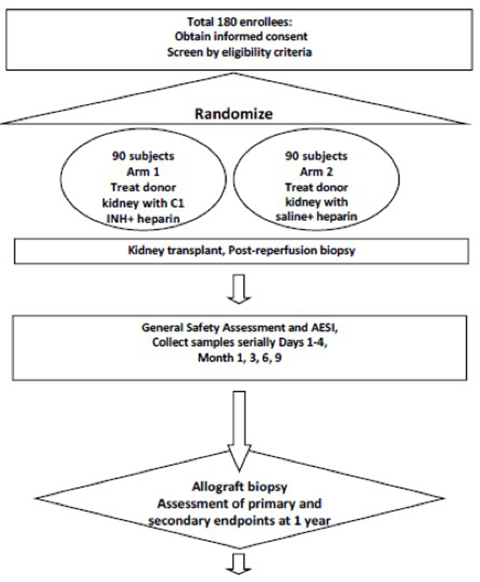
Schematic design of the IMPROVE trial.
Additional Trials
My laboratory functions as a mechanistic core laboratory for NIH-funded clinical trials testing roles of tocilizumab (anti IL-6R) in heart transplant recipients and separately in lung transplant recipients, and testing safety and efficacy of belatacept in heart transplant recipients.
Contact the Heeger Lab
Steven Spielberg Building
8723 Alden Dr., Room 322
Los Angeles, CA 90048