Research Areas
The research focus of the Ramani Lab is to identify molecular factors that contribute to liver diseases, ALD, MASLD, liver fibrosis and liver cancer, and assess their viability as therapeutic targets.
Role of A-Kinase Anchoring Proteins in Liver Fibrosis
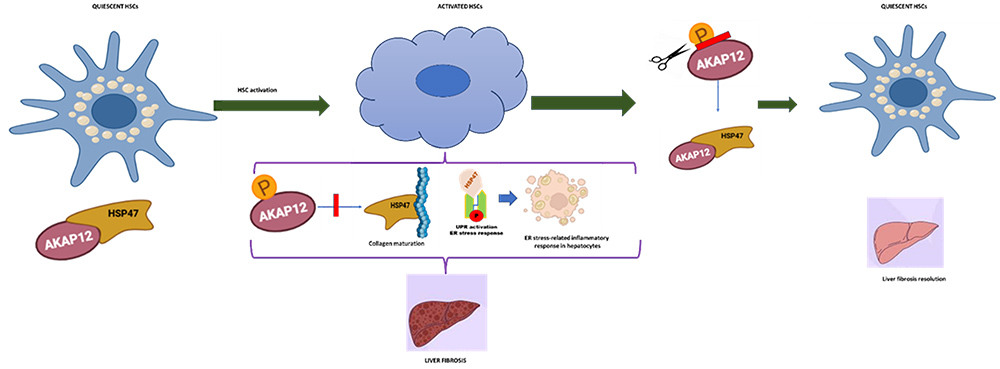
Hepatic stellate cell (HSC) activation is a hallmark of liver fibrosis, characterized by an increased secretion of extracellular matrix components, collagens and smooth muscle actin that disrupts hepatic architecture. We identified the scaffolding protein called A-kinase anchoring protein 12 (AKAP12) as an interacting partner of the collagen chaperone heat shock protein 47 (HSP47) in quiescent HSCs. During activation, AKAP12 undergoes site-specific phosphorylation that disrupts this interaction, thereby liberating HSP47 to accelerate collagen maturation. In animal models, gene editing of these activation-responsive phospho-sites preserved the AKAP12-HSP47 scaffold, which in turn regulated collagen production and attenuated HSC activation, inflammation and ER stress mediated by HSP47s interaction with unfolding protein response machinery (UPR). Consequently, inhibiting AKAP12 phosphorylation exerts potent anti-fibrotic effects, suggesting a novel therapeutic target for liver fibrosis.
AKAP Scaffolding Functions in Alcohol-Associated Liver Disease
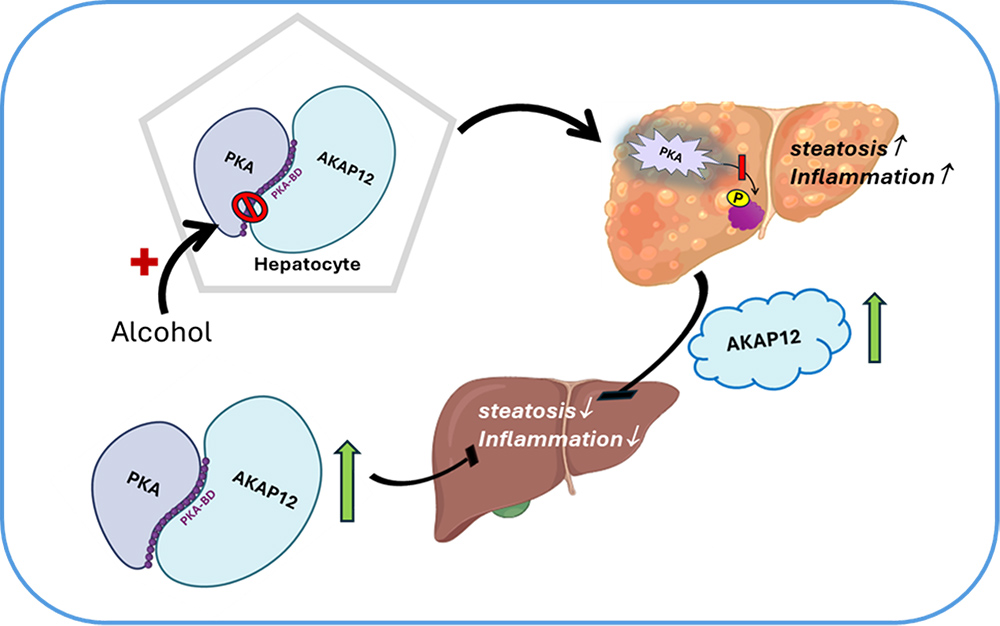
AKAP12 is known to interact with the PKA-binding domain (PKA-BD) of protein kinase A or PKA, increasing PKA-dependent phosphorylation of its substrates, sustaining PKA activation in hepatocytes. This regulates lipogenesis and promotes fatty acid oxidation. Inhibition of the AKAP12-PKA scaffold by alcohol or by editing the PKA-BD suppresses PKA activation in the liver, altering the activity of PKA substrates that are controlled by PKA-dependent phosphorylation. Loss of the AKAP12-PKA scaffold thereby alters the lipogenic program and increases steatosis and inflammation. Increased AKAP12 expression in the liver can overcome steatosis in an alcohol-damaged liver and is associated with decreased inflammatory signaling in hepatocytes. Therapeutic strategies to sustain AKAP12-PKA scaffold such as peptide mimetics could be a potential approach targeting alcohol-associated liver disease. An important scaffolding function of AKAP12 controlled by β2-adrenergic signaling involves activation of lipolytic signaling in the liver. AKAP12 can promote hepatic lipolysis that is disrupted by alcohol exposure. Studies are underway to evaluate these molecular mechanisms in the context of AKAP12’s role as a mediator of lipid homeostasis.
LARP1 as a Therapeutic Target for MASLD Progression
In collaboration with Shelly Lu, MD, the Women's Guild Chair in Gastroenterology, we investigated how the S-adenosylmethionine (SAMe)-synthesizing enzyme Mat1a suppresses tumorigenesis in mice. Continuing this collaborative project, our work focuses on defining the contribution of SAMe deficiency to the progression from metabolic dysfunction-associated steatotic liver disease (MASLD) and metabolic dysfunction-associated steatohepatitis (MASH) to hepatocellular carcinoma (HCC). We have demonstrated that dysregulation of the protein translation machinery in SAMe-deficient systems promotes HCC development in the context of MASLD. In recent work, we identified La-related protein 1 or LARP1 as a key translational regulator whose phosphorylation drives oncogenic mRNA translation, as well as enhanced growth and migratory capacity of liver cancer cells. Furthermore, through integrated phosphoproteomic and proteomic analyses, we showed that the MASLD phenotype in the SAMe-deficient, Mat1a-knockout mouse model is characterized by a global hyperphosphorylation signature, mitochondrial dysfunction and increased peroxisomal β-oxidation. We discovered that LARP1 might regulate these pathways through its ability to alter translation of associated targets. Ongoing studies are aimed at identifying and functionally characterizing molecular drivers emerging from this proteomic analysis that contribute to MASH-to-HCC progression.
Contact the Ramani Lab
Davis Building, Suites 2093 (Office) and 3094C (Laboratory)
110 George Burns Road
Los Angeles, CA 90048