January 2023 Case
Clinical History
A toddler that was recently admitted at an outside hospital for a large abdominal mass, confirmed to be of hepatic origin on CT and MR scans. There was no evidence of extrahepatic disease on imaging. Serum AFP was markedly elevated (>500,000) at the time of diagnosis. A chemotherapy regimen was started consisting of cisplatin, 5FU, vincristine, and doxorubicin. The tumor showed shrinkage and the patient's AFP levels went down markedly after chemotherapy. The patient subsequently underwent left hepatectomy.
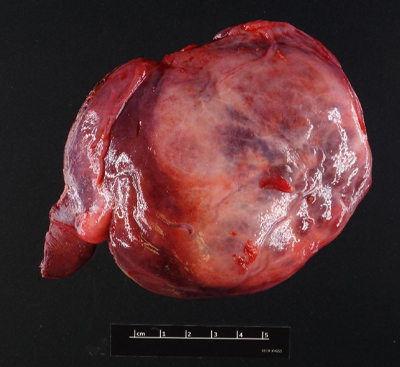
Figure 1. (Gross photo) Gross photo shows large mass within the liver (right) and adjacent uninvolved liver parenchyma (left).
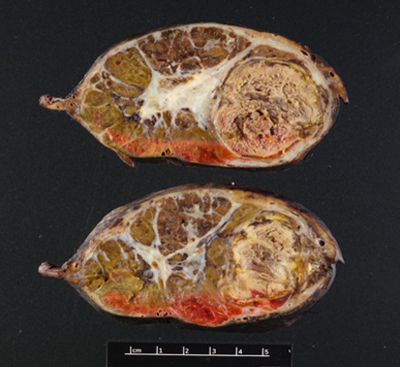
Figure 2. (Gross Photo, Cross Section) Cross sections show multilobulated tumor with areas of necrosis and central scarring.
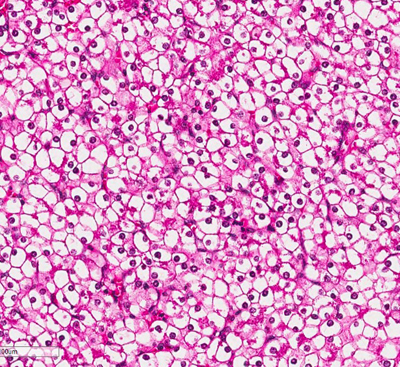
Figure 3. (Microscopic Photo, 20x) The fetal pattern of the tumor is composed of uniform cells resembling normal fetal hepatocytes. The cells are small with round centrally located nuclei, abundant clear to pink cytoplasm, and have distinct membranes.
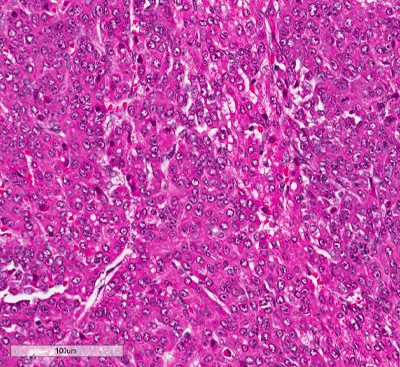
Figure 4. (Microscopic Photo, 20x) The embryonal pattern consists of more primitive appearing cells in acini and sheets. These cells are small and angulated. The nuclei are larger than those of the fetal component nuclei and have coarse chromatin, scant cytoplasm, and indistinct membranes.

Figure 5. (Microscopic Photo, 10x) In addition to the epithelial component, there are osteoid-like elements consistent with a mesenchymal component to the tumor.
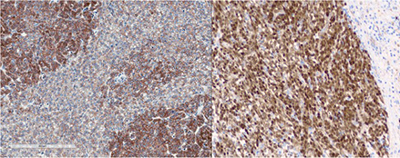
Figure 6. (Microscopic Photo, 10x) IHC for Beta-Catenin shows tumor with nuclear staining and membranous staining.
Diagnosis
Hepatoblastoma, mixed epithelial and mesenchymal type.
Discussion
Hepatoblastoma is a rare primary liver malignancy often diagnosed in the first three years of life with an incidence of 1.8 per million [1]. It is the most common liver malignancy in children [2,3]. Though most cases are sporadic, hepatoblastoma has a known association with Beckwith-Wiedemann Syndrome (11p15.5) and Familial Adenomatous Polyposis (5q21), and case reports link hepatoblastoma to many other genetic syndromes including Trisomy 18, Fragile X, Prader-Willi, and Noonan syndrome [4]. Prematurity, and very low- or high-birth weight have also been associated [5]. At the genomic level, extra copies of chromosome 2, 8, or 20, loss of chromosome 4 or 18, and translocations involving 1q12 or 1q24 have been reported in hepatoblastoma [5]. Treatments include surgical resection and chemotherapy.
Hepatoblastoma is classified according to histologic type. The two major histological categories are epithelial and mesenchymal, and tumors may be entirely epithelial or have mixed epithelial and mesenchymal components. Epithelial components are classified into subtypes or patterns including: fetal, embryonal, small cell undifferentiated, macrotrabecular, pleomorphic and cholangioblastic Fetal pattern is further subclassified into well-differentiated/mitotically inactive and crowded/mitotically active based on mitotic activity and cytologic features. Epithelial patterns may occur alone or in combination and may transition from one pattern to another on a spectrum. Fetal with or without admixed embryonal patterns are most frequently encountered. The presence of a mesenchymal component in addition to an epithelial component of any subtype defines a mixed epithelial and mesenchymal tumor. The mesenchymal component most commonly includes osteoid, though other elements including cartilage, skeletal muscle, fat and spindle cell components can rarely be present. If neural or neuroectodermal differentiating is present, the tumor is classified as having teratoid features. Though mesenchymal components are common in the post-chemotherapy setting, it is in fact an intrinsic part of the tumor and not an effect or therapy [1, 2, 3].
Gradually, the therapeutic approach for hepatoblastoma has become more individualized with better patient stratification based upon clinical and pathologic features. Although initially all hepatoblastomas were treated in the same way, later at least two new patient categories emerged [6]. The PRETEXT system aims for staging and risk stratification of liver tumors at diagnosis. Standard-risk hepatoblastomas, are tumors which are entirely limited to the liver and associated with elevated AFP levels (>100 ng/mL). High-risk hepatoblastomas, are tumors which are metastatic or involving the whole liver or have extrahepatic/vascular extension or are associated with low AFP or a combination of these factors. Histologic features may also impact prognosis. The presence of pure fetal, well-differentiated/mitotically inactive pattern is associated with good prognosis. The entire tumor must be examined to classify as pure fetal, well-differentiated/mitotically inactive. Other patterns including small cell undifferentiated pattern may be associated with poor prognosis; however, further larger scale and more comprehensive studies are needed to fully assess this. Vascular invasion is also associated with worse prognosis [2,3,]. Treatment involves surgery, neoadjuvant or adjuvant chemotherapy, and or orthotopic liver transplant.
References
1. Sumazin P, Chen Y, Treviño L, Sarabia S, Hampton O, Patel K, et al. Genomic analysis of hepatoblastoma identifies distinct molecular and prognostic subgroups. Hepatology. 2017;65(1):104-21. Doi:10.1002/hep.28888.
2. Cho SJ. Pediatric Liver Tumors: Updates in Classification. Surg Pathol Clin. 2020 Dec;13(4):601-623. doi: 10.1016/j.path.2020.09.002. PMID: 33183723.
3. Ranganathan S, Lopez-Terrada D, Alaggio R. Hepatoblastoma and Pediatric Hepatocellular Carcinoma: An Update. Pediatr Dev Pathol. 2020 Mar-Apr;23(2):79-95. doi: 10.1177/1093526619875228. Epub 2019 Sep 25. PMID: 31554479.
4. Venkatramani R, Spector L, Georgieff M, Tomlinson G, Krailo M, Malogolowkin M, et al. Congenital abnormalities and hepatoblastoma: a report from the Children’s Oncology Group (COG) and the Utah Population Database (UPDB). Am J Med Genet A. 2014;164A(9):2250-5. Doi:10.1002/ajmg.a.36638.
5. Tomlinson G, and Kappler R. Genetics and epigenetics of hepatoblastoma. Pediatr. Blood Cancer. 2012;59: 785-92. Doi:10.1002/pbc.24213.
6. Morgan Auld F, Sergi CM. Surgical Pathology Diagnostic Pitfalls of Hepatoblastoma. Int J Surg Pathol. 2022 Aug;30(5):480-491.